By melding two microscopy methods and a computational tool, researchers can quickly and precisely quantify neuronal connections in individual animals, according to a new study. The technique could make it faster to map the connectomes of autism mouse models and track how mutations in autism-associated genes rewire neural circuits.
A human neuron has thousands of synaptic connections, which light-based microscopy lacks the resolution to detect. Electron microscopy can resolve neuronal links in exquisite detail — theoretically down to 0.12 nanometers, a length slightly shorter than a carbon-carbon chemical bond — but the process is slow and laborious. In one study, it took about three years to section and image a single fly brain with a scanning electron microscope.
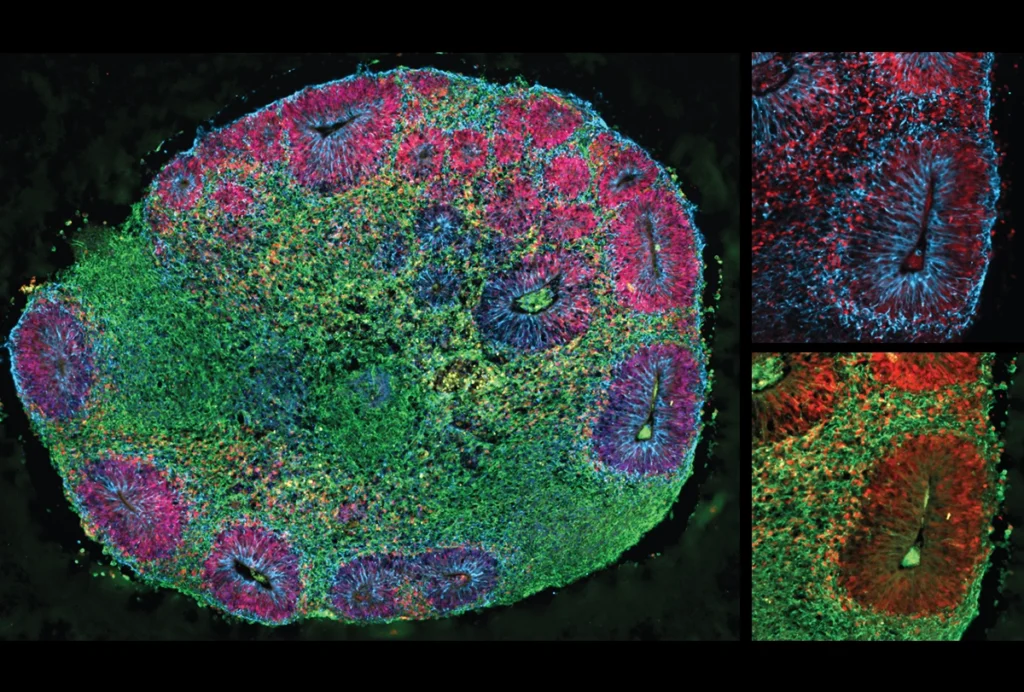
The new approach leans on tissue-expansion methods to achieve a similar resolution but at the speed of light-based microscopy. The team first labels specific proteins in a chunk of brain — such as those at presynaptic sites or gap junctions, which mediate electrical signals between cells. They then swell the tissue to eight times its normal volume and take a series of 2D pictures using lattice light-sheet microscopy, which illuminates an entire plane of tissue at once.
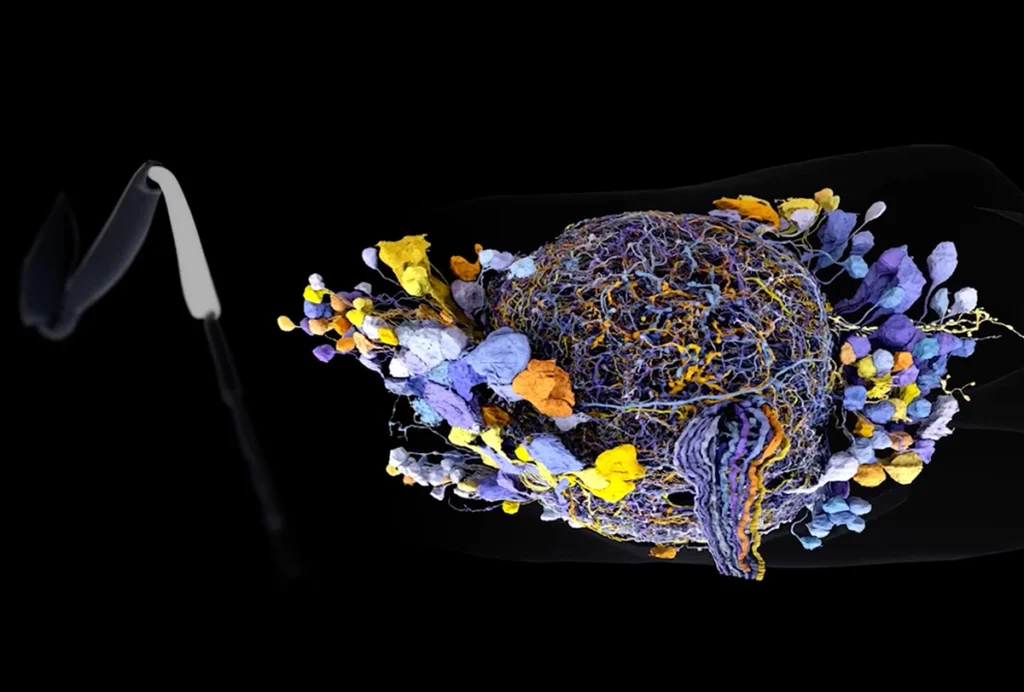
Finally, they stitch the images together into a 3D video. A freely available machine-learning software tool can then automatically locate and quantify the number and size of neuronal connections and export these data to a table for later analyses. The entire protocol takes about one week and is well suited for use with mice, fruit flies, nematodes and other organisms, says study investigator Joshua Lillvis, research scientist at the Howard Hughes Medical Institute’s Janelia Research Campus in Ashburn, Virginia.
“We can do actual quantification of synapses at a structural level, and we can see if they change based on experience or neural dysfunction across species,” Lillvis says. “There’s a lot of opportunity here, and we’re excited to see what people do with it.”
A
The new study, which was published in eLife in October, addresses both issues. To confirm that light-sheet microscopy and tissue expansion produce similar results to electron microscopy, Lillvis and his colleagues extracted the optic lobe from fruit flies and used an antibody to label a scaffolding protein called Bruchpilot at presynaptic sites. Electron microscopy images indicated that each neuron had, on average, 207 presynaptic sites, comparable to that recorded using the light-sheet method.
Solving the file-size issue demanded a different tact. Lillvis’ team modified a free software tool called VVD Viewer to support large image files and stitch them together into 3D space. They also developed a convolutional neural network, trained on images of different synapses, to automatically detect synapses and segment individual neurons in the images.
“The major contribution here is in the computational pipeline and analysis,” says Limor Freifeld, assistant professor of biomedical engineering at the Technion – Israel Institute of Technology in Haifa, who was not involved in the work. “Also, the fact that they take these large datasets and, in an automatic manner, reliably segment the neurons and synapses and quantify the data — that’s significant.”
T
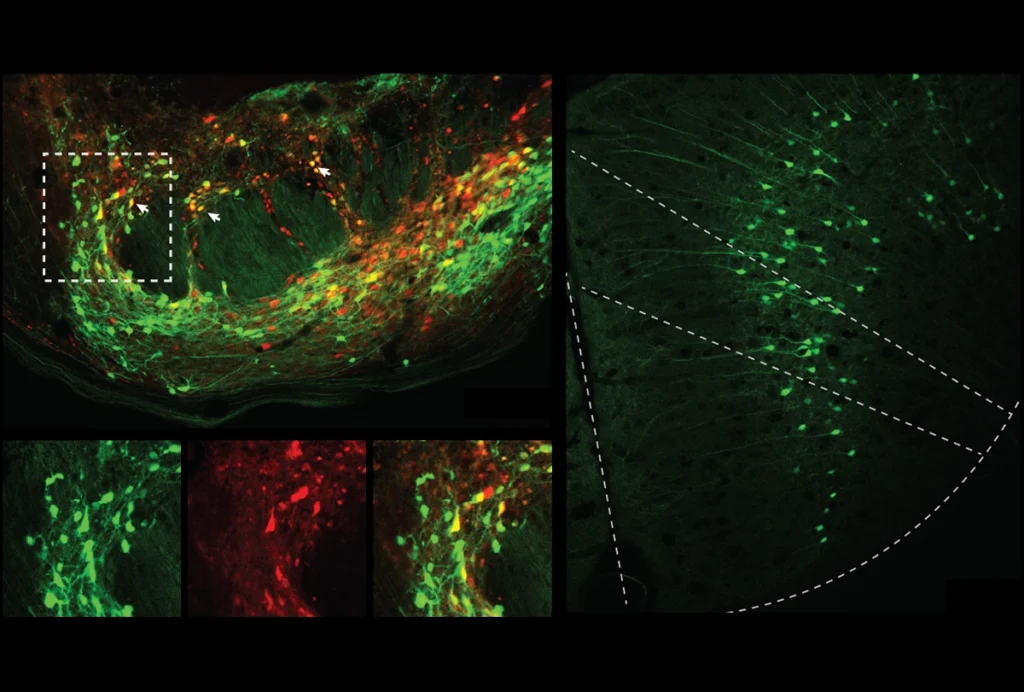
As a proof of concept, Lillvis labeled two types of neurons in the fly brain, called pC2l and pIP10, that are involved in male courtship. Each fly sings using different sounds — pulses and hums — to woo a female, a behavior triggered upon activation of the pC2l neurons. The pIP10 neurons are both necessary and sufficient for flies to make pulse sounds, and they are also thought to determine which specific song each fly sings. Scientists knew that these two types of neurons were physically connected, but not how strongly they interact.