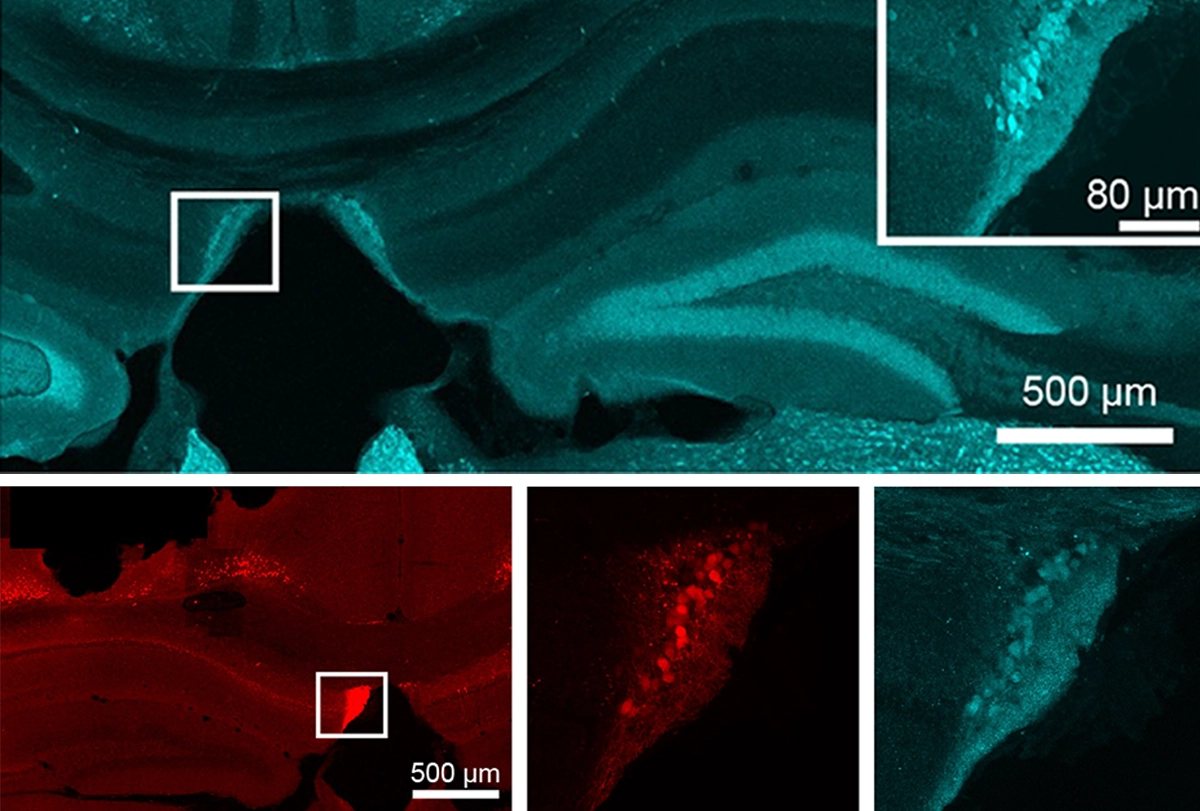
Five years ago, while working to develop a tool to label neurons active during seizures in mice, Quynh Anh Nguyen noticed something she had not seen before.
“There was a particular region in the brain that seemed to light up really prominently,” she says.
Nguyen, assistant professor of pharmacology at Vanderbilt University, had induced seizures in the animals by injecting kainic acid into the hippocampus—a common strategy to model temporal lobe epilepsy. The condition often involves hyperactivity in the anterior and middle regions of the hippocampus, but Nguyen’s mice also showed the activation in a tiny posterior part of the hippocampus that she was not familiar with.
Nguyen brought the data to her then-supervisor Ivan Soltesz, professor of neurosciences and neurosurgery at Stanford University. Together they realized that these neurons were in an area called the fasciola cinereum—a subregion of the hippocampus so understudied, Soltesz says, that when Nguyen first asked him what it was, he had “no idea.”
Despite the subregion’s obscurity, it looks to be an important and previously overlooked contributor to epilepsy in people who do not respond to anti-seizure medications or tissue ablation in the hippocampus, Nguyen and her colleagues say. Fasciola cinereum neurons were active during seizures in six people with drug-resistant epilepsy, the team reported in April.
Inhibiting fasciola cinereum neurons optogenetically shortens the duration of seizures in model mice, that study also shows. And seizures decreased in frequency in one participant who underwent ablation to remove the subregion—hinting that the fasciola cinereum could serve as a novel treatment target.
The “compelling” work involves a “clever set of experiments,” says Robert Hunt, associate professor of anatomy and neurobiology and director of the Epilepsy Research Center at University of California, Irvine, who was not involved in the study. But whether the hard-to-reach region can feasibly be targeted in other people remains to be seen, he adds.
T
o translate the results from mice to people, study investigator Ryan Jamiolkowski, a neurosurgery resident at Stanford, decided to try putting stereoelectroencephalography (sEEG) electrodes—used to localize the source of hyperactivity in a brain—in the fasciola cinereum region of the six people with drug-resistant epilepsy.Typically, the portions of the hippocampus that are monitored and ablated do not include this region, Soltesz says. And because the fasciola cinereum is so difficult to reach, Nguyen says, “you really have to have an almost dedicated electrode just to try to target the FC.”
Despite that challenge, all six people showed seizure activity in the fasciola cinereum, the researchers found. One participant had already undergone surgery to ablate the anterior hippocampus and amygdala but was still experiencing a high volume of seizures; about one third of people with medial temporal lobe epilepsy who undergo ablation surgery do not experience seizure relief. Ablating the fasciola cinereum, however, decreased the participant’s seizure frequency by 83 percent after 18 months.
“We are not saying that the seizures are starting there,” Soltesz says, but the area is “involved,” and hopefully the findings will one day help more people.
For now, however promising the results, “this is where kind of the science meets reality,” Hunt says. “It gets really narrow towards the posterior end of the hippocampus, and it sits right between the thalamus and the occipital cortex. There’s a lot of visual areas in there, and white matter tracts,” he says, making the region a risky area to ablate using current technology.
This study is only one of two from the past two decades focused on the fasciola cinereum, according to PubMed. The other, published in 2022, defined the anatomy of the subregion and explored the physiological characteristics of the cells in rats.
The fasciola cinereum is a “very well-defined independent area” with “unique connectivity” between it, the entorhinal cortex and the dentate gyrus, says 2022 study investigator Inah Lee, chair of the brain and cognitive sciences department at Seoul National University. It is important for contextual memory and has place cells, but “compared to other place cells in other structures, like CA1, they are not very stable,” Lee says.
Nguyen, who launched her own lab this year, says she plans to focus her attention on a range of basic questions about the fasciola cinereum—including, for example, its cellular composition, which cell populations engage during seizures and how these cells connect to the seizure network.
“There’s a lot of really big questions that we’re hopefully gonna tackle and answer,” she says.