Mutations between genes, long overlooked, may be key in autism
Spontaneous mutations that occur between genes may be as important in autism as those within genes.
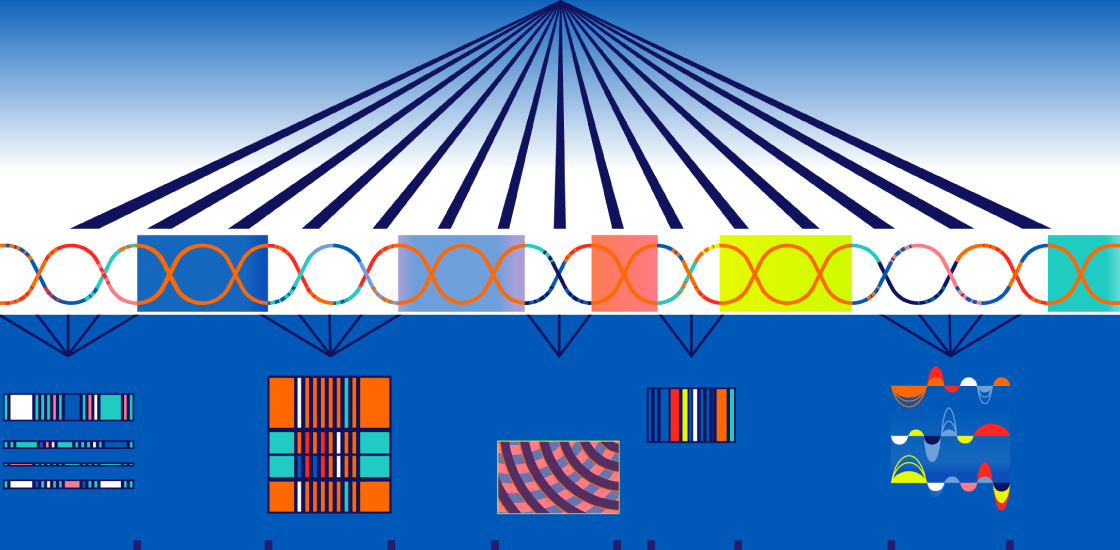
Spontaneous mutations that occur between genes are as important in autism as those within genes, a new study suggests1.
The study, published today in Nature Genetics, is the first to look at the impact of these ‘noncoding’ mutations across the whole genomes of autistic people.
Many teams over the past three years have sequenced the DNA of autistic people both within and between genes. Yet sorting through the hundreds of thousands of mutations between genes had seemed nearly impossible because researchers know so little about these genetic segments.
The new study overcomes this challenge by using a machine-learning approach. The researchers created an algorithm that predicts whether a particular noncoding mutation alters any gene’s expression. It assigns each mutation a score based on how likely it is to do so — and to be harmful.
“The unique approach here is that instead of just counting mutations, we’re using the deep-learning-based frameworks to look at their regulatory impacts,” says co-lead author Olga Troyanskaya, professor of integrative genomics at Princeton University in New Jersey. “All mutations are not created equal, and all effects are not created equal.” (Troyanskaya also holds a position at the Simons Foundation, Spectrum’s parent organization.)
A strength of the study is that it looks at spontaneous mutations across the entire genome, experts say.
Previous efforts to analyze noncoding mutations focused on certain regions, often those closest to genes, notes Xin He, assistant professor of genetics at the University of Chicago, who was not involved in the study.
“Here, just genome-wide, you see a clear signal,” He says. “That’s a very impressive result.”
Binding influence:
Troyanskaya’s team and her colleagues analyzed 7,097 whole genomes from 1,790 families that have one child with autism but unaffected parents and at least one unaffected sibling. They found tens of thousands of spontaneous mutations in the autistic children, but about the same in the unaffected siblings. The algorithm they created predicts the likelihood that a mutation disrupts a genomic region that controls gene expression.
The team then searched the Human Gene Mutation Database to check whether any of the mutations have known links to a medical condition or tend to appear in controls. They pulled together all of this information to generate an impact score for each mutation.
Noncoding mutations in children with autism have greater impact scores, on average, than those in their unaffected siblings, the researchers found.
Overall, the analysis suggests, such mutations account for autism in 4.3 percent of children with the condition; by comparison, harmful mutations within genes account for 5.4 percent.
The team also assessed the mutations’ effect on messenger RNA (mRNA), the intermediary between gene and protein. In autistic children, they found, mutations that disrupt mRNA have a greater impact than those that affect DNA.
“This is an interesting thing to follow up,” says Lucia Peixoto, assistant professor of biomedical sciences at Washington State University Spokane, who was not involved in the study. “I don’t think a lot of people in autism are actually looking at RNA processing as an important pathway.”
The genes closest to the high-impact noncoding mutations in autistic children tend to be expressed in brain tissue, the researchers found.
“It’s nice to see multiple lines of evidence support this idea that one way to get autism and other neurodevelopmental disorders is by affecting the noncoding regulatory elements in the developing brain,” says Jeffrey Barrett, chief scientific officer of Genomics plc, a biotechnology company based in Oxford, England, who was not involved with the study.
Neighboring genes also tend to be involved in processes altered in autism, such as neuronal signaling or gene regulation — the same pathways that coding mutations disrupt.
The team also assessed the effects of high-impact mutations on gene expression in cultured cells. In most cases, they found, the mutations seem to alter the expression of a gene.
The algorithm, called DeepSEA, is available online. Troyanskaya and her colleagues are applying their method to the whole genomes of individuals with other conditions, such as congenital heart disease.
- Zhou J. et al. Nat. Genetics Epub ahead of print (2019) Abstract
This article was republished in Scientific American.
Recommended reading

Building an autism research registry: Q&A with Tony Charman
CNTNAP2 variants; trait trajectories; sensory reactivity
Brain organoid size matches intensity of social problems in autistic people
Explore more from The Transmitter
Cerebellar circuit may convert expected pain relief into real thing