Some questions about neurons, such as how they give rise to behavior, are tricky to answer when those cells are embedded within their natural milieu.
“Is residence in a nervous system sufficient to allow synapses to form?” says Kristin Baldwin, professor of genetics and development at Columbia University. “Are synapses that we can see sufficient to allow communication? And is synaptic communication sufficient to actually endow an animal with a set of behaviors that would be appropriate for it?”
The best way to answer those questions is to put the cells in a new environment where their extrinsic and intrinsic influences can be teased apart, says Xin Jin, assistant professor of neuroscience at the Scripps Research Institute.
For a long time, Jin says, that new environment was the unnatural setting of a petri dish. But two new studies that make use of chimeric mice—animals with both mouse and rat cells in their brain—point to another option: One demonstrates how rat stem cells can restore a mouse’s ability to smell, whereas the other shows how rat stem cells can give rise to a forebrain in mice that would otherwise lack one. The studies were published last month in Cell. Because rat brains are larger than mouse brains and develop at a different rate, the chimeras enable researchers to probe the competing forces of a cell’s intrinsic programming and its external environment.
The work opens up doors for new research and the ability to explore the origins of species-specific cellular behaviors, says Jin, who was not involved in either study.
“It’s sort of a fundamental ‘nature versus nurture,’” says Baldwin, who led one of the new studies.
B
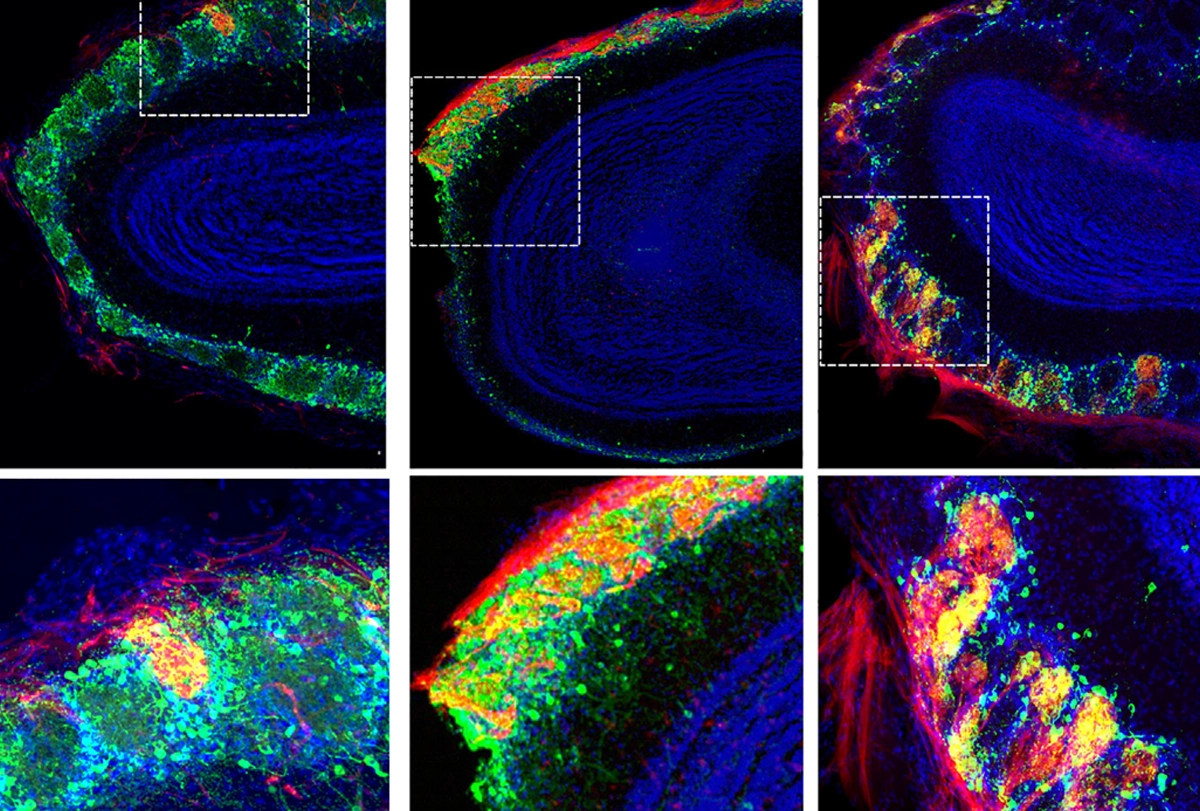
aldwin and her colleagues engineered mice with olfactory sensory neurons—the cells in the nose that detect odors—that had been either genetically killed or, through expression of a neurotoxin, synaptically silenced. They then injected both groups of developing mouse blastocysts with embryonic stem cells from rats—a technically challenging feat, Jin says.The rat cells successfully integrated themselves into olfactory circuits whether the mouse’s olfactory sensory neurons were absent or silent, Baldwin and her colleagues found. But animals with silenced olfactory neurons could not find a buried cookie by scent alone, whereas those that lacked those cells could put those new rat neurons to use.
The distinction may point to some competition between remaining mouse cells and the introduced rat cells, Baldwin says. “That alone gives us a new handle on what are the pathways and molecules and cellular responses that are consistent with a functional rescue, and those maybe that are not,” she adds.
Rat stem cells can also grow to replace entire structures missing in a mouse brain, according to the second study. Researchers injected the cells into the blastocysts of mice engineered to lack the gene HESX1, which is essential for the development of the forebrain. On their own, mice lacking HESX1 have no forebrain and do not survive past birth. But when the rat cells are present, the team found, they grow alongside mouse cells and help form a forebrain—and the mice survive and behave typically.