For autism mouse models, gender matters
The mutation that leads to Angelman syndrome may affect the brains of female mice more severely than those of male mice.
The mutation that leads to Angelman syndrome may affect the brains of female mice more severely than those of male mice, according to unpublished results presented today at the 2015 Society for Neuroscience annual meeting in Chicago.
The findings are part of a large project that is rigorously characterizing changes to the brain structure of dozens of mouse models of autism. By examining both sexes, the researchers are capturing nuances that might be missed by focusing exclusively on males, says Jacob Ellegood, a research associate at the Mouse Imaging Centre, in Toronto, who presented the findings at a poster session.
The mice in the study are missing the UBE3A gene on the chromosome they inherited from their mothers. Because the paternal gene copy is always silenced, the mice make no UBE3A protein. In people, mutations on the maternal copy of UBE3A lead to Angelman syndrome, an autism-related developmental disorder characterized by intellectual disability, epilepsy and a happy demeanor.
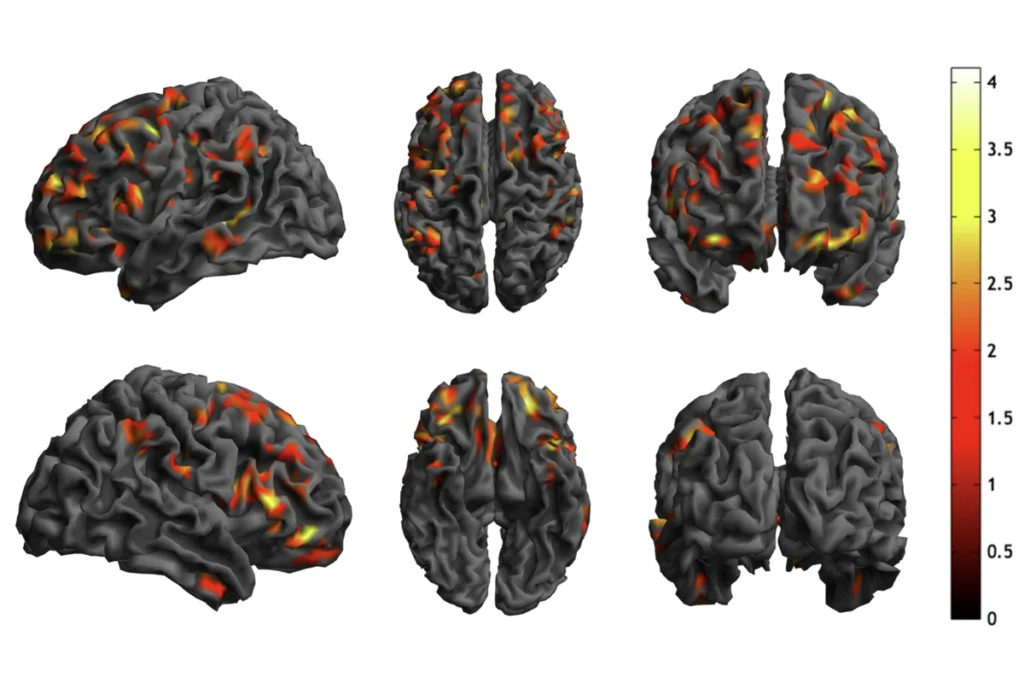
The researchers scanned the brains of 10 male and 8 female mice with the mutation, and 11 male and 10 female otherwise identical controls. They compared the size of 62 brain segments across mutant mice and controls. They overlay these size differences on a detailed atlas of the mouse brain to determine which brain structures are affected.
Shrinking brains:
A number of brain regions are smaller in the male and female mutant mice than in controls. One of the most strikingly diminutive regions is the thalamus, a sensory and motor relay station. One region of the hippocampus and part of the striatum — which function in memory and reward, respectively — are also smaller in mutant mice.
For all of these brain regions, the size decreases are more pronounced in females than in males. The one exception is a portion of the cerebellum, which coordinates movement. It shows greater shrinkage in males than in females.
In the second phase of the project, Ellegood and his colleagues aim to associate these brain changes with characteristics in the mice. Each examined brain is from a mouse that took a battery of behavioral tests, starting at birth.
Both male and female UBE3A mutant mice have delays opening their eyes at birth, a sign of general cognitive delay. As pups, they also make fewer and shorter calls to their mothers. And as juveniles, the male mutant mice — but not the females — are less active than controls.
So far, the researchers see greater brain changes in the female mice but greater behavioral deficits in the males. But this incongruity may be due to the fact that, so far, they have been looking at changes in entire brain regions, Ellegood says.
Ellegood has both imaging and behavioral data on each mouse, so he will now be able to match behavioral test scores for individual mice with changes in each of the 62 segments.
“I’m hoping that we’ll see even more differences between males and females when we start correlating with behavior,” he says.
Ellegood and his colleagues plan to repeat this approach with other mouse models of autism, including mice lacking the fragile X syndrome gene. His team is also building on a project they published in 2013 in which they looked for patterns of alterations in the brains of 26 different autism-related mouse models.
“If I were to redo that paper today, there would be well over 50 brains,” he says. “And they just keep coming.”
For more reports from the 2015 Society for Neuroscience annual meeting, please click here.
Recommended reading
Okur-Chung neurodevelopmental syndrome; excess CSF; autistic girls

New catalog charts familial ties from autism to 90 other conditions
Explore more from The Transmitter

Karen Adolph explains how we develop our ability to move through the world
Microglia’s pruning function called into question