Jump-starting growth signaling reverses microcephaly in autism mouse model
A genetic therapy and an existing drug both restore typical brain size in mice missing DYRK1A, a top autism candidate gene, in the cerebral cortex, a new study shows. The animals typically have smaller brains than controls.
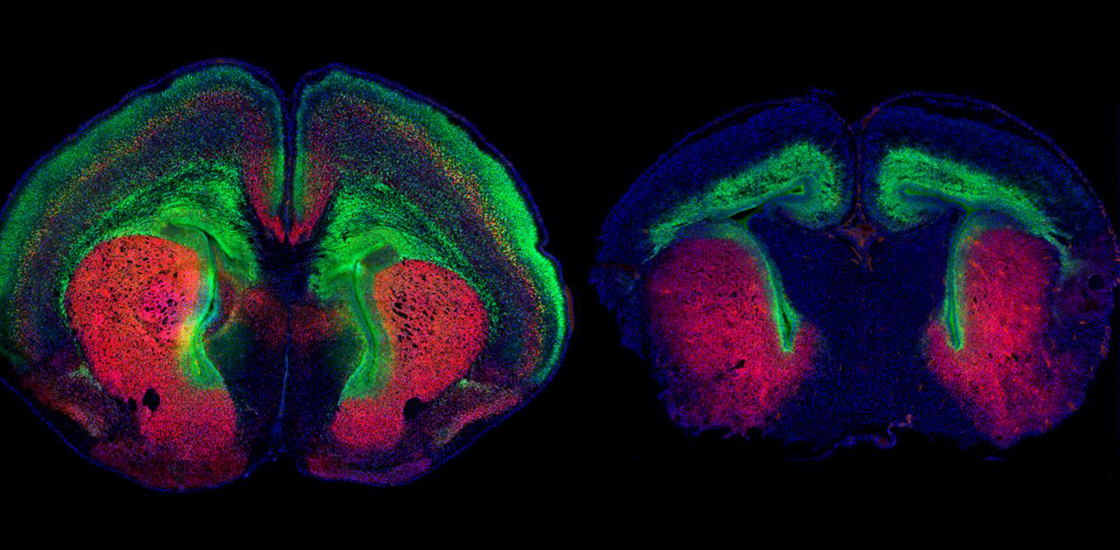
Mice missing DYRK1A, a top autism candidate gene, in the cerebral cortex have unusually small brains, a new study shows. But using a genetic approach or an existing drug to restore cell growth pathways in the mice rescues their brain size.
Many people with a mutated copy of DYRK1A have autism and intellectual disability or developmental delay. They also have a smaller-than-average head, or microcephaly.
The DYRK1A mice show traits reminiscent of autism, such as a reduced preference for socializing with other mice. Two factors largely account for the animals’ small brains, the new work shows: cells that are smaller and more tightly packed than usual, and increased apoptosis, or cell death.
“At birth, these animals have a normal number of cells in the brain, but they actually seem to be smaller. But then neurons are progressively lost as the animals develop, and that seems to be happening by apoptosis,” says lead researcher Damon Page, associate professor of neuroscience at Scripps Research in Jupiter, Florida.
Treating the mice with insulin-like growth factor 1 (IGF-1), approved by the U.S. Food and Drug Administration (FDA) to treat short stature, normalizes both their behavioral traits and physical brain changes, Page and his colleagues found. The study was published earlier this month in Biological Psychiatry.
“One of the most exciting strengths of this work is that the authors are able to rescue the neural phenotypes pharmacologically, and one of the tested approaches is already FDA-approved,” says Summer Thyme, assistant professor of neurobiology at the University of Alabama at Birmingham, who was not involved in the study.
Growth pathways:
Page’s team looked at two types of DYRK1A mice: one with a functional copy of the gene in the cerebral cortex, the other with none. The mice missing both copies of the gene did not survive beyond their first day of life.
Both types of mice had a smaller brain and cerebral cortex at birth than controls, attributes that persisted into adulthood for those with one functional copy of DYRK1A. The mice also had physical brain features like those seen in people with the mutation, including enlarged fluid-filled chambers called ventricles and undergrowth of the corpus callosum, the bundle of nerve fibers that unites the brain’s two hemispheres.
But both types of mice had the typical number of neurons in the cerebral cortex, according to a cell count done by isotropic fractionation. Therefore, their smaller brain size is likely due to smaller cells and closer spacing, Page says.
Apoptosis or decreased cell proliferation in the mice before birth, neither of which the team assessed, could also contribute to microcephaly in the animals, says Noelle Dwyer, associate professor of cell biology at the University of Virginia in Charlottesville, who was not involved in the study.
“It is surprising that they’re saying there were not effects on proliferation or apoptosis, that it’s all about cell size and density,” she says. “It could well be, I just wonder if we can be completely confident about that.”
But Page says his team saw no difference in total cell number at birth, which is inconsistent with the idea that altered apoptosis or cell proliferation played a role.
Cell staining of tissue from adult mice did reveal increased apoptosis. These mice also showed a lowered preference for interacting with other mice.
Several proteins involved in signaling pathways that promote cell growth, including brain-derived neurotrophic factor (BDNF), mTOR and IGF-1, were suppressed in the DYRK1A mice, mass spectrometry showed.
The pathways that involve these molecules also play a role in other genetic forms of autism — a link that makes this study’s findings particularly exciting, Thyme says.
Reverse effects:
Whereas DYRK1A activates these pathways, another autism-linked gene, PTEN, suppresses them. PTEN mutations often lead to brain overgrowth rather than microcephaly.
So Page and his team suspected that suppressing PTEN might be therapeutic for mice missing DYRK1A. Indeed, breeding the DYRK1A mice with mice that carry a mutated copy of PTEN yielded pups whose brains and behavior closely resembled those of controls.
In another effort to overcome DYRK1A deficiency in mice missing one copy of the gene, Page’s team administered a form of IGF-1 to the animals just after birth. Within days, this too restored the mice’s cerebral cortex to the usual size. It also restored structural complexity to cultured neurons from the mice.
Previous work has shown that IGF-1 can reduce autism traits in mice with mutations in SHANK3, another gene linked to autism, and clinical trials of the compound are in progress for people with SHANK3 mutations.
Page’s team is testing IGF-1 treatment on DYRK1A mice at different stages of development to determine whether treatment later in life can restore brain size.
Explore more from The Transmitter
The illusion of AI consciousness: Lessons from human unconscious processing
‘Push-pull’ recipe for neural wiring used in multiple brain regions