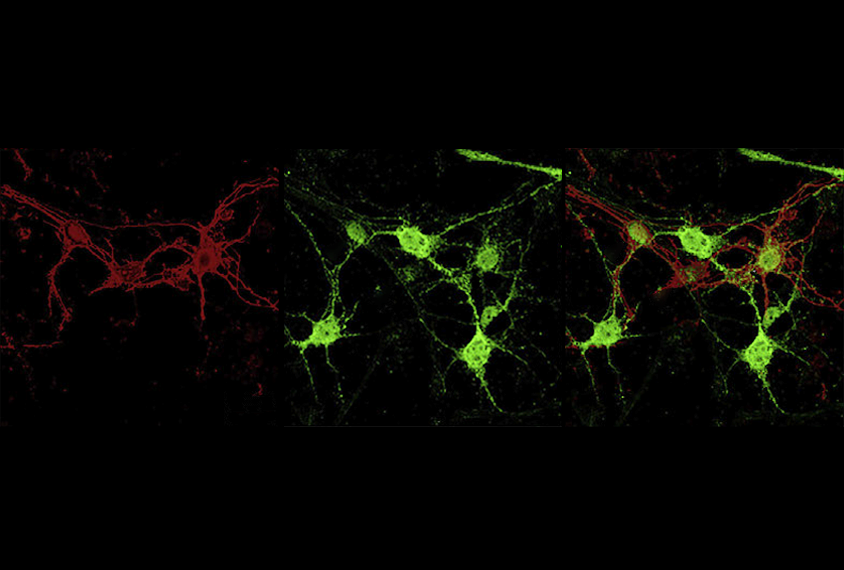
Some alternate RNA versions, or isoforms, don’t have a fighting chance. They contain segments that mark them for immediate degradation — meaning they produce no protein, even when the gene is functional. This mechanism enables cells to regulate their protein expression by changing up how a gene’s RNA isoforms are pieced together.
The autism-linked gene SYNGAP1 is one of many genes that make this type of nonproductive isoform, according to a new study. An antisense oligonucleotide (ASO) that switches SYNGAP1’s nonproductive form to a productive one increases protein expression in brain organoids, the work also shows.
“It’s a proof of principle,” says Gavin Rumbaugh, professor of neuroscience at UF Scripps Biomedical Research in Jupiter, Florida, who was not involved in the work. The findings help explain how SYNGAP1 is regulated throughout development and point to a mechanism that may one day help treat SYNGAP1 deficiency, a genetic condition that can lead to autism and intellectual disability, Rumbaugh says.
An approved treatment for spinal muscular atrophy, a genetic condition, harnesses a similar “splice-switching” approach, as does an experimental treatment for Dravet syndrome, a rare form of epilepsy.
Although the SYNGAP1 results are exciting, they are only a first step, says lead investigator Xiaochang Zhang, assistant professor of human genetics at the University of Chicago in Illinois.
“We think it’s a promising target,” Zhang says. But before this approach can move to the clinic, “there are still a lot of things to do.”
Z
hang and his colleagues identified the genetic code that flags nonproductive SYNGAP1 isoforms for degradation, as well as the proteins, called PTBPs, that regulate which isoforms get made.The nonproductive SYNGAP1 isoform is conserved between mice and humans, Zhang’s team confirmed. Some people who have autism or intellectual disability harbor SYNGAP1 mutations that result in the degradation code being included in isoforms too frequently, the researchers found.
The SYNGAP1 protein is most prevalent at neuronal synapses, where it is thought to facilitate learning and memory. Mice deficient in SYNGAP1 protein have neurons with weakened synaptic plasticity, and their cells require more synaptic input for excitation, past studies have shown.
Removing the degradation code from SYNGAP1 via CRISPR engineering increases levels of the protein in mice, the team found. Performing the same genetic deletion in mice that carry one deficient copy of SYNGAP1 restores the animals’ synaptic plasticity and neuronal excitability.
The researchers also identified an ASO that suppresses the production of the nonproductive SYNGAP1 isoform and increases production of one that makes SYNGAP1 protein. Applying this ASO to stem cells obtained from people who carry a SYNGAP1 mutation that limits the gene’s expression increases the amount of productive mRNA isoforms in the cells, the team found. And treating cortical organoids with the same ASO boosts their SYNGAP1 protein production. The findings were published this month in Neuron.
T
he results are further evidence that “you can take advantage of the body’s natural mechanisms to regulate gene expression,” says Joseph Gleeson, professor of neurosciences at the University of California, San Diego, who was not involved in the work. The approach, he says, could lead to “a wave of therapeutics.” (Gleeson is a consultant for the biotechnology company Ionis, which develops ASOs to treat genetic conditions, and chief medical officer of the n-lorem Foundation, which freely provides experimental antisense drugs to people with “nano-rare” diseases.)But the new study has its limitations, Gleeson says. The researchers “show that they can increase the amount of protein that’s made” using their ASO, but they do not confirm that the ASO rescues the atypical electrophysiological properties seen in neurons from SYNGAP1-deficient animals, he says. “They brought it only so far.”
It may be that the ASO doesn’t boost SYNGAP1 high enough or affect the brain early enough to improve neuronal function, Gleeson says, in which case it would be premature to call this a potential avenue for therapy.
Zhang and his colleagues agree that the ASO needs to be tested further before this can be thought of as a viable treatment. “We want to be very conservative in terms of interpretation,” he says.
Treating SYNGAP1-deficient mice with the ASO is an important next step, Zhang says, as is determining which brain regions the small molecule can reach and at what point in development the treatment would be effective.
“I’m very convinced that this splicing is a real mechanism that regulates SYNGAP1 protein,” Rumbaugh says. “And not only that — I’m convinced that if someone could figure out a way to make this happen in humans with SYNGAP1 haploinsufficiency, that it might actually improve their phenotypes.” But figuring that out, he adds, is no simple task.