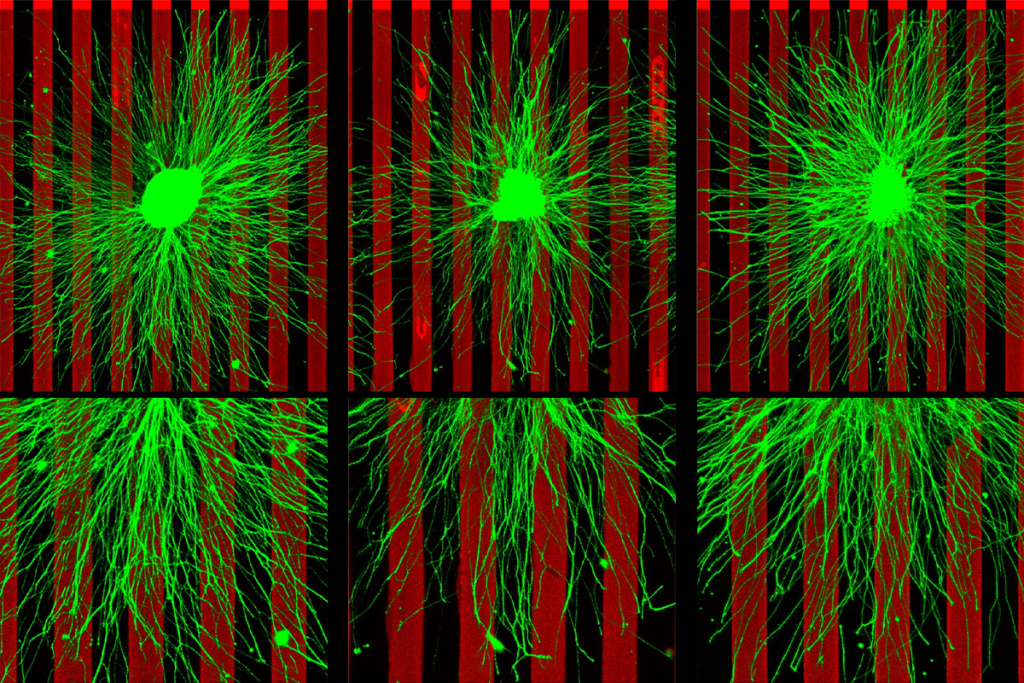
Two unapproved drugs that boost levels of the neurotransmitter serotonin significantly increase sociability in six autism mouse models, according to a new study.
The findings build on a decade of work that suggests the release of serotonin in the nucleus accumbens, part of the brain’s social-reward system, is important for social interactions and promoting sociability, says lead researcher Robert Malenka, professor of psychiatry and behavioral sciences at Stanford University in California. Artificially stimulating the release of serotonin in the nucleus accumbens in mice with an autism-linked mutation lessens their reluctance to socialize with other mice, a 2018 study from Malenka’s team showed.
In the new work, the team used MDMA, also known as ecstasy, and an experimental serotonin agonist called CP-94,253 to stimulate serotonin circuitry in multiple autism mouse models. Both drugs restored the animals’ interest in socialization to levels near those of their wildtype peers.
“It really is not what I expected,” Malenka says. “I thought it might work in one or two models but not the other ones. But we kept doing model after model, and it kept working.” Malenka is a founder and scientific advisory board member at Maplight Therapeutics, a biotech company recruiting participants for a phase II trial of a different serotonin agonist for autism.
The findings could lead to medications to ease social difficulties in people with autism, the researchers say, though other experts urge caution.
The behavioral tests used in the study measure only narrow dimensions of social interest in mice, and it will be important to evaluate whether these drugs truly increase social function in people, says Jeremy Veenstra-VanderWeele, professor of psychiatry at Columbia University, who was not involved in the study. “These tests are oriented toward social interest or social engagement. Many autistic people are quite interested in other people but still struggle socially, and a drug that increases social interest may not help them.”
Targeting sociability:
In the new study, Malenka and his colleagues tested each drug in mice with a mutation in the chromosomal region 16p11.2 or in the genes CNTNAP2 or FMR1, all of which are linked to autism. They also tested the drugs in mice exposed to the epilepsy drug valproic acid in the womb, which is associated with autism. Additionally, they tested CP-94,253 in mice with mutations in the genes ACTL6B or ARID1B, both of which have autism ties.
The team chose these models in part because each one involves a different biological pathway but yields the same behavior — a reticence about socializing, compared with wildtype mice. The work appeared 8 July in Neuropsychopharmacology.
The two drugs in the study have different mechanisms and also differ from the class of drugs called selective serotonin reuptake inhibitors (SSRIs), which can treat co-occurring conditions such as depression and anxiety but do not appear to affect core autism traits. Whereas CP-94,253 activates a set of serotonin receptors, MDMA stimulates the release of serotonin. SSRIs, on the other hand, slow the rate at which serotonin is recycled.
All three boost serotonin levels, but CP-94,253 and MDMA do so within minutes or hours, whereas SSRIs work in a matter of weeks.
Clinical value:
The range of mouse models that responded to the study drugs lends support to the idea that disparate autism-linked genetic mutations may converge on the same pathways and that treating those pathways will improve functioning in people, says Elizabeth Berry-Kravis, professor of child neurology at Rush University Medical Center in Chicago, Illinois, who was not involved in this work. “It’s nice to see some support for that concept.”
But previous drug trials have shown great promise in mice and then failed to produce similar results in people, she says. “I’ve become a very healthy skeptic of things that fix the mouse.”
Future clinical trials should test the potential drugs in people screened to show the greatest response, she says. Screening could ensure that participants share a genetic marker, brainwave signature or social trait that helps unify the group and gives researchers something to measure to see an effect.
This subtyping is great in theory, Malenka says. “In practice, especially for a small biotech, it’s basically impossible.”
In the meantime, serotonin agonists approved for use in people already exist. For example, triptans, commonly used to treat or prevent migraines, seem relatively safe in people, so the idea that this class of drugs might have potential for autism is exciting, Veenstra-VanderWeele says.
“To my knowledge, people who take those medicines do not report increased social engagement,” he says, “but they are usually taking these medicines for debilitating headaches, so social engagement may not be the top concern.”