Early changes in brain immunity may underlie sex bias in autism
How the brain’s immune cells develop in ‘germ-free’ mice might help explain why autism is more prevalent in boys than girls.
How the brain’s immune cells, or microglia, develop in ‘germ-free’ mice depends on the sex of the mice, according to a new study1. If the findings hold up in people, this sex difference may help to explain why four times as many boys as girls are diagnosed with autism.
The mice are raised in sterile conditions, and they — as well as their mothers — lack a microbiome, the body’s natural community of microbes. As such, the mice represent an extreme version of a scenario in which there is some environmental perturbation during gestation, such as a maternal infection.
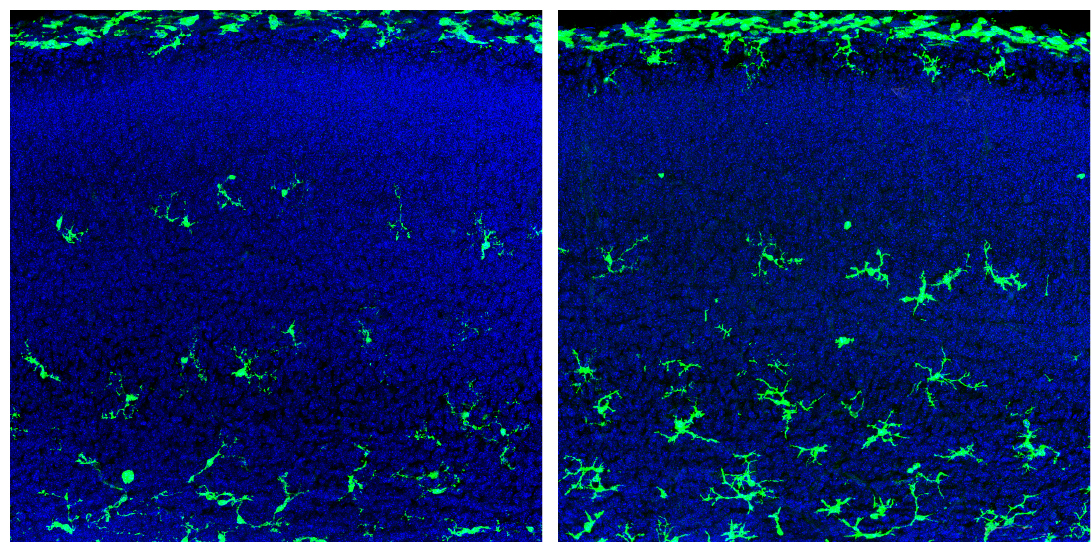
The study reveals that the microglia increase in density and show significant changes in gene expression only in male fetal mice; in adulthood, only females display large-scale changes in density and gene expression in their microglia.
“There’s a critical window during development where only males are very sensitive to the lack of a microbiome,” says co-lead researcher Florent Ginhoux, a principal investigator at the Singapore Immunology Network.
Microglia clear away debris and defend against pathogens. They also help to sculpt the brain’s wiring by wolfing down certain synapses, the connections between neurons. Defects in microglia have been linked to autism.
“Many of us think that autism is largely prenatal in origin, so the idea that the [microglia in] males are more vulnerable early in life really fits with that,” says Staci Bilbo, director of research at the Lurie Center for Autism at Massachusetts General Hospital in Boston, who was not involved in the study.
Brain’s defenders:
In 2015, researchers in Germany showed that mice lacking a microbiome have defective microglia as adults, hinting that the microbiome may influence microglia2.
In the new work, Ginhoux and his colleagues explored whether the body’s microbes influence microglia function during fetal development, and whether this influence shifts with age and by sex. They isolated microglia from typical male and female fetal mice at five time points, as well as from adult mice.
“We wanted to know if microglia could constitute sort of an entry point for environmental signals in regulating the wiring of [brain] circuits,” says co-lead investigator Sonia Garel, professor of brain development and plasticity at the Institut de Biologie de l’École Normale Supérieure in Paris.
They found only slight differences in gene expression in the microglia of male and female fetal mice. Both sets express some of the genes that adult microglia rely on to probe their surroundings and detect pathogens.
This finding suggests that fetal microglia are also capable of sensing — and responding to — environmental changes. The study appeared 21 December in Cell.
Role reversal:
The sex differences are stark in microglia from the pups of germ-free mice, however. Compared with controls, germ-free male fetal mice show altered expression of roughly 1,200 genes — including a decrease in the levels of immune system genes. By contrast, germ-free female fetuses show differences in only about 20 genes.
This sex difference is reversed in adult mice. Only 26 genes are altered in adult germ-free males, compared with 433 in females. Like germ-free male fetuses, the adult females also show a decrease in the expression of immune system genes.
The germ-free conditions also trigger an increase in the density of microglia compared with controls. This effect is stronger in the germ-free male fetuses than in the females, and the opposite is true in the adults.
“Something about the microbiome is changing the local environment in the brain differently in males versus females,” says Margaret McCarthy, professor and chair of pharmacology at the University of Maryland, who was not involved in the study. “How that is coming about is just completely unknown, and very intriguing and exciting to think about.”
The differences may relate to sex-specific hormone levels, which begin to diverge near the end of embryonic development in mice, she says.
When the researchers treated typical mice with an antibiotic for a week to partially wipe out their microbiomes, they saw only mild sex differences in the gene expression profiles of microglia, and no effect on density.
These findings suggest that the sex differences seen in the germ-free adult mice are the consequence of long-term depletion of microbes.
Fetal position:
As a first step toward extrapolating the results to people, the researchers isolated microglia from six male and four female human fetuses. As in mice, the fetal microglia show few sex differences in gene expression.
These findings are at odds with an unpublished analysis of human brain tissue, which suggests that typical male fetuses express higher levels of microglia genes than do females.
The discrepancy may be because that study analyzed whole brain tissue, which contains a mixture of cell types rather than only microglia, says Donna Werling, a postdoctoral fellow in Stephan Sanders’ and Matthew State’s labs at the University of California, San Francisco. Werling is an investigator on the unpublished study but was not involved in the new work.
The new results highlight the importance of including female mice in research, something that all studies funded by the U.S. National Institutes of Health have been required to do since late 2014.
“I think people have been really reluctant to include both [sexes] because they think, ‘Well, it’s twice the work,’” Bilbo says. “But one of the most important takeaways of the work is that you’re missing so much of the biology by not including females.”
Scientists should explore whether the microbiome also influences particular brain circuits or behaviors in sex-specific ways, some experts say.
“If we could identify the factors that make the female microglia less vulnerable to immunological events, this may open future avenues for therapeutic targets,” says Jaclyn Schwarz, assistant professor of psychological and brain sciences at the University of Delaware in Newark, who was not involved in the study.
In the meantime, the researchers plan to study how microglia respond to more realistic prenatal complications tied to autism risk, such as maternal infection and preterm birth.
References:
Explore more from The Transmitter
Neuroscience conference policy draws confusion, apology

Funding for animal research alternatives reaches ‘inflection point’
