A previously unrecognized population of fibroblasts seals off the base of the choroid plexus—the network of blood vessels and cerebrospinal-fluid-producing epithelial cells that line the ventricles—from the cerebrospinal fluid (CSF) and the rest of the brain, a new study in mice shows.
The newly identified barrier provides an added layer of protection that is distinct from the well-known blood-brain barrier and the one that the epithelial cells form between the blood and the CSF.
The findings help settle a long-standing debate about whether there was a blind spot in the choroid plexus that gave the periphery access into the brain, says Britta Engelhardt, professor of immunobiology at the University of Bern, who was not involved in the work. “Some [scientists] speculated that there is a leak, like an opening, a secret window into the brain, and others said, ‘No, there must be a barrier that we have overlooked.’ And it’s very obvious now.”
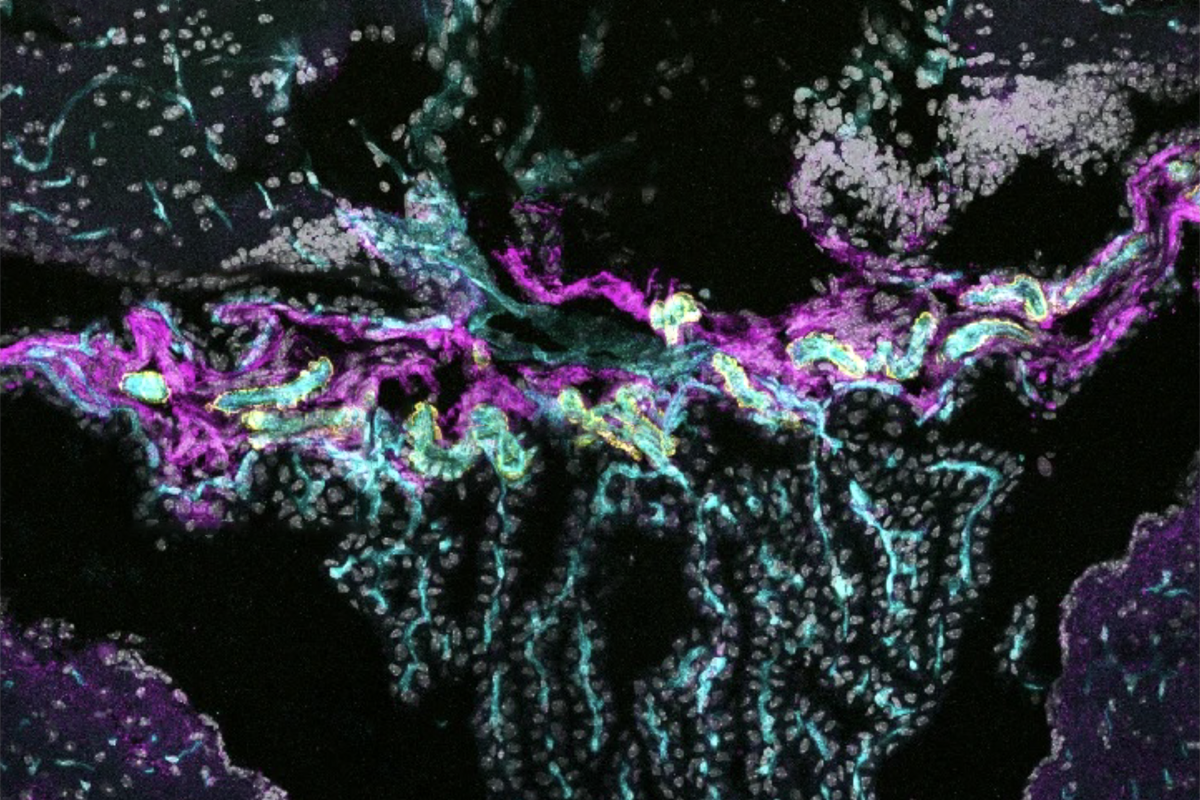
Fibroblasts at the base of the choroid plexus, connected by adherens and tight junction proteins, cluster together around blood vessels and form a sealed barrier in mice, the researchers found. This structure represents a crucial component of compartmentalization in the choroid plexus, Engelhardt says. The cells were also present in human postmortem brain samples.
Similar to other barriers, the seal becomes leaky in response to inflammation triggered by lipopolysaccharide, a component of the bacterial cell wall, and it may coordinate immune cell crossing from the blood into the brain, the study also showed. The work was published in February in Nature Neuroscience.
The results suggest that neuroscientists who study how immune cells interact with brain barriers may have been looking in the wrong place, says study investigator Roosmarijn Vandenbroucke, professor of biomedical molecular biology at Ghent University. “That changes our focus a bit from not only looking at the blood-CSF barrier but also […] at the base barrier as a potential, maybe more preferred,” point of entry for immune cells during inflammation.
And this novel structure can now be studied alongside other barriers, Engelhardt says. “We have now discovered that there is a connection from the outer leptomeninges all the way to the choroid plexus, and the question is: Where along this pathway are the barriers?” she says. “This [study] is now setting the stage.”
V
She failed to find any interesting signals, she says. “To be honest, the results were a bit disappointing.” But when she looked more closely at the fibroblasts’ expression, she noticed they segregated into two distinct populations. One of these showed barrier-like properties, including expression of the tight junction protein CLDN11, which piqued Vandenbroucke’s interest.
These CLDN11-expressing fibroblasts resided at the base of the choroid plexus and clustered together, forming tight junctions that sealed the region, the researchers found through electron microscopy. The barrier was also functional, tracer experiments revealed. Tracer injected into the blood failed to access the CSF, whereas tracer injected into the ventricles remained there without crossing into the choroid plexus.
The combination of these findings shows that “it’s a true, true barrier,” Engelhardt says.
The fibroblasts, which the group named “base barrier cells,” express many of the same genes as the cells that make up the meningeal arachnoid barrier, the study found. And they come from the meninges, lineage tracing shows, which “highlights how interconnected brain borders can be from very early in development,” says Carmen Ruiz de Almodóvar, professor of neurovascular cell biology at the University of Bonn, who was not involved with the study.
T
Humans, too, have cells at the base of the choroid plexus that express CLDN11 and present honeycomb-like patterns typical of tight junctions, the study found, suggesting the barrier is conserved across species.
Next, it will be important to understand how these newly identified cells connect to other tissues and fit into the rest of the barriers around the brain, and how these might synergistically coordinate to regulate brain function, Ruiz de Almodóvar says. “Because at the end, they all aim for protecting the brain and allowing the brain to receive the molecules that [it] needs.”