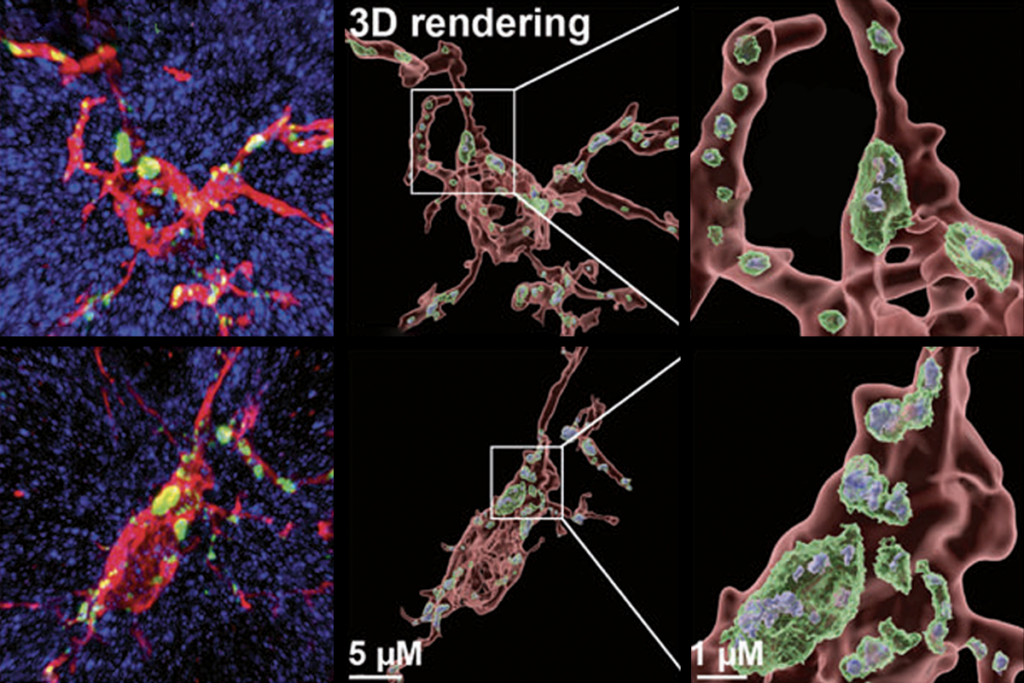
New drug rescues function in fragile X syndrome
A new drug appears to relieve symptoms of fragile X syndrome by blocking the over-production of a key protein in a subset of people with the disorder, according to a 6 January study in Science Translational Medicine.
A new drug appears to relieve symptoms of fragile X syndrome by blocking the over-production of a key protein in a subset of people with the disorder, according to a 6 January study in Science Translational Medicine1.
Fragile X syndrome, an inherited form of mental retardation with features of autism, is caused by a mutation in the FMR1 gene. Some individuals with fragile X have the full mutation, which shuts down production of the protein, FMRP, needed to form healthy connections between neurons. Others have a partial mutation, which spares some FMRP production.
FMRP acts as a brake on components of the mGluR5 pathway, which run riot in individuals with the full mutation, leading to severe symptoms of the disorder. The new study shows that the drug AFQ056, made by Swiss pharmaceutical giant Novartis, blocks production of mGluR5, lessening the severity of symptoms in individuals with the full mutation. Individuals with partial mutations show more variable results.
Earlier studies have shown that mGluR5 antagonists rescue FMRP production in animal and cell culture models. But this is the first study to validate the hypothesis that blocking the mGluR5 system has a positive effect in people with fragile X syndrome.
“This study gives us hope that mGluR blockers will be a good treatment that will at least partially reverse symptoms of fragile X syndrome,” says Elizabeth Berry-Kravis, co-director of the Molecular Diagnostics Laboratory at Rush University Medical Center in Chicago. Berry-Kravis was a member of the study design team, providing clinical advice on outcome measures that could be used to assess the drug’s efficacy.
Model theory:
Researchers first proposed the mGluR theory in 20042. Loss of FMRP at synapses, the junction between neurons, allows mGluR signaling to kick into overdrive, they suggested, leading to symptoms of fragile X by preventing the synaptic changes necessary for learning and memory.
“Since that initial presentation, everyone has been very excited about the idea that you could block the mGluR system at whatever level — maybe by blocking the receptor or maybe by blocking the signaling pathway — and that might be a targeted treatment to partially reverse symptoms of fragile X,” says Berry-Kravis.
Over the past several years, a number of studies have provided support for the hypothesis in various model systems, including mice, Drosophila and cultured neurons. And Novartis is one of many companies banking on mGluR blockers.
Until now, however, it wasn’t clear whether results in other models would translate to humans. “One never knows what’s going to happen in the jump from mice to humans because the human brain wires over a more prolonged period of time,” points out Berry-Kravis.
Stephen Warren, chair of human genetics at Emory University in Atlanta, is one of the researchers who first proposed the mGluR theory. He says the new study is “hopeful, but not definitive,” partly due to the small sample size: 30 participants.
Warren says he is disappointed that the researchers did not provide information on the number of DNA repeats in the FMR1 genes of participants, because the size of the mutation plays a role in how much FMRP protein is made. “I was surprised that they didn’t show that data, because it could clarify their findings quite a bit,” he says.
The researchers did not directly measure FMRP levels, inferring protein levels from mRNA production. “That’s a weakness of the study,” Berry-Kravis admits, and one that will be addressed in a trial under way aiming to validate the results in a larger sample.
Mosaic effects:
In the study, 16 participants with fragile X syndrome received AFQ056 and 14 received placebo over a 30-day period. On day 20, researchers evaluated the drug’s efficacy by administering the Aberrant Behavior Checklist, a behavior rating scale that measures five domains, including irritability, stereotypies and hyperactivity3. They found no statistically significant improvement. However, when they used another outcome measure, the Repetitive Behavior Scale4, clear indications of improvement emerged.
These effects were magnified when the researchers divided study participants into two subgroups, based on whether the FMR1 promoter is fully or partially methylated. Methylation — the addition of a methyl group to molecules — shuts down protein expression. In the study, individuals whose FMR1 genes are fully methylated showed significant improvement after treatment with AFQ056 compared with the placebo group.
“The changes were quite significant — in the range of 20 to 45 points on the Aberrant Behavior Checklist,” says Baltazar Gomez-Mancilla, lead investigator on the study. “But we still don’t entirely understand whether there is a correlation.”
Because individuals with full methylation are more severely affected by the disorder, their improvement is likely to be more dramatic, the researchers note. By contrast, individuals whose FMR1 genes are only partially methylated — methylation mosaics — show greater variability in response to treatment.
Because the researchers don’t provide data on either size mosaicism — variability in the number of unmethylated pre-mutation alleles — or FMRP levels, however, it’s difficult to determine how much FMRP the individuals in the partially methylated group produce and whether that affects treatment outcome.
Assessing outcomes is particularly difficult in studies on fragile X syndrome because none of the testing instruments were developed specifically for the disorder, notes Berry-Kravis. The researchers chose the Aberrant Behavior Checklist because it has been used in drug approval studies for Risperdal and Abilify, drugs used to treat autism.
She is participating in an effort spearheaded by researchers at the M.I.N.D. Institute at the University of California, Davis, to validate outcome measures for studies of fragile X syndrome. “It’s complicated, because we are trying to determine the optimal outcome measure at the same time [that] we are trying to figure out if the drug works,” she says.