Worms and zebrafish missing both copies of the gene CHD7 have a disrupted cellular signaling pathway, a dearth of inhibitory neurons and behavioral challenges. The stimulant drug ephedrine restores all of these changes, according to a new study.
People with a mutated copy of CHD7 often have CHARGE syndrome, a condition that can cause heart and eye defects, stunted growth, attention deficit hyperactivity disorder and autism.
Zebrafish lacking both copies of the gene are hyperactive at night, unlike controls and fish missing just one copy, the new study shows.
“This hinted to us that something might be going wrong in the circuitry of the brain,” says lead researcher Kessen Patten, associate professor of genetics and neurodegenerative disease at the Institut national de la recherche scientifique in Quebec, Canada.
Patten and his colleagues found that the fish show dysregulation along the MAPK/ERK signaling pathway, which is involved in cell growth and implicated in other autism-related neurodevelopmental conditions. The animals also have fewer interneurons that produce gamma-aminobutyric acid (GABA), which dampens excitatory signals in the brain. A common theory of autism holds that the condition is caused by an excitatory signaling imbalance.
It’s not totally clear how relevant the model zebrafish are to people with CHD7 mutations, says Carlos Parras, a researcher at the Paris Brain Institute in France, who was not involved in the study. People with CHARGE syndrome generally have only one altered copy of the gene, whereas the fish have two. But because treating the fish soon after hatching restored atypical brain signaling and behavior, he says, it is possible that administering a drug during brain development might similarly help people.
Cascading effects:
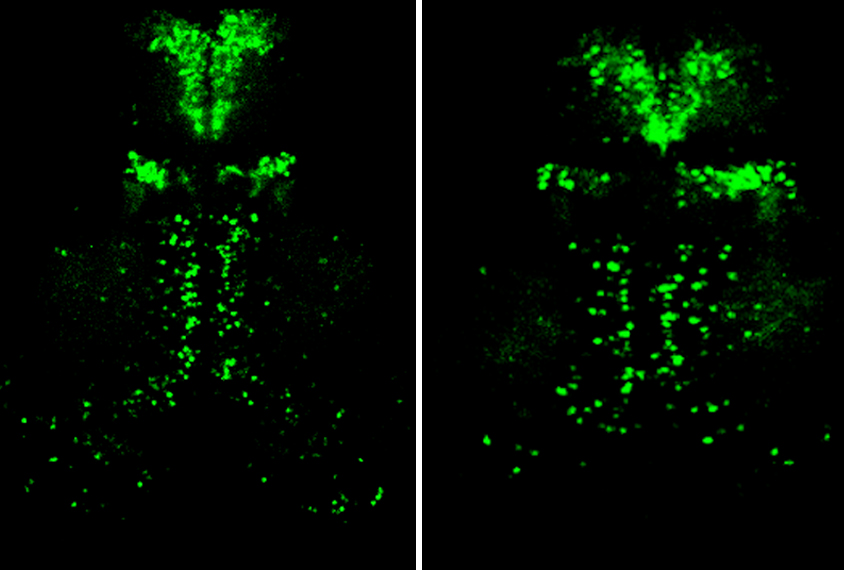
Using a fluorescent tag, Patten and his colleagues found significantly fewer GABAergic interneurons in the brains of larval zebrafish lacking CHD7 than in those of controls. Cell staining further revealed that precursors to these cells, called progenitor neurons, did not effectively differentiate.
Because larval and newly hatched fish have so few cells, the analysis seems “very preliminary,” Parras says. “To me, it is not clear how the loss of CHD7 is mediating this effect.”
But Patten and his team propose that the loss of CHD7 initiates a cascade of effects: It suppresses the expression of PAQR3B, a gene that inhibits the MAPK/ERK cell signaling pathway, which in turn leads to fewer GABAergic interneurons and hyperactivity in the animals.
RNA sequencing showed that the CHD7 fish had more than 1,000 dysregulated genes, including many involved in MAPK signaling; PAQR3B was the most dysregulated of all. A similar analysis of cells from people with a mutated copy of CHD7 also showed PAQR3B dysregulation.
Fluorescent reporter genes in Caenorhabditis elegans worms also revealed that the loss of CHD7 disrupted GABA signaling networks. The model worms showed impaired swimming abilities compared with controls.
The work appeared in April in EMBO Reports.
Stimulant treatment:
Boosting the expression of PAQR3B in the model zebrafish larvae by injecting them with PAQR3B messenger RNA improved, but did not fully restore, GABA cell development. And treating the fish with two drugs that activate GABA receptors restored their nighttime activity to levels seen in controls.
But to find the best target along the cascade of effects, the team turned to the model C. elegans to screen 3,850 drugs — 49 of which improved the worms’ ability to swim. Patten’s team tested the three compounds that had the strongest effects in the worms on the CHD7 fish. They found that one, the stimulant ephedrine, significantly restored GABA neuron function and suppressed hyperactivity in the fish.
This result supports the idea that “the GABAergic defect was responsible, at least in part, for the behavioral defects in CHD7 mutant animals,” Parras says.
Ephedrine is a stimulant that functions similarly to amphetamine, a frontline treatment for attention deficit hyperactivity disorder. Both increase levels of GABA in people. Something similar could be happening in this model, Patten says, but his results do not suggest that people with CHARGE syndrome should take ephedrine, which can cause cardiovascular problems at higher doses.
“There’s a lot more to be done before we can go in the clinic and give it to patients,” he says.