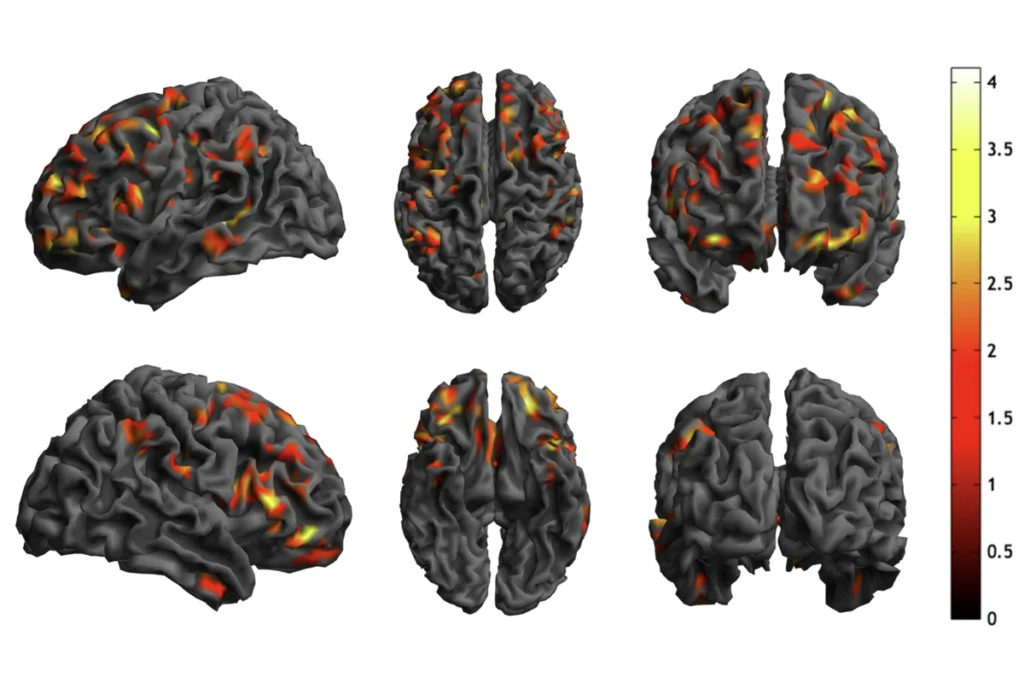
Top autism gene may rewire brain by altering expression of other genes
The autism gene TBR1 controls the expression of several other candidate genes that govern the architecture of the brain’s outer section.
The autism gene TBR1 controls the expression of several other candidate genes that govern the architecture of the cerebral cortex, the brain’s outer section, a new study suggests1.
TBR1 is among a select set of genes with strong ties to autism. The new findings explain that connection: Mutations in TBR1 may disrupt gene expression in a way that alters brain circuits.
When the gene is disabled in layer 6 — the deepest layer — of the cerebral cortex of mice at birth, the neurons there do not wire up the way they typically do. They also have unusual gene-expression patterns.
“When major neurons like this are confused in terms of the information they are gathering and sending, that’s a huge problem for normal circuits,” says lead investigator John Rubenstein, professor of psychiatry at the University of California, San Francisco.
Restoring the expression of WNT7b, one of the genes under TBR1’s control, reverses some of the problems with the circuits, providing a clue to a therapy.
Network news:
In 2010, the team reported that mutant mice lacking TBR1 show altered gene expression in layers 5 and 6 of the cerebral cortex2.
In the new work, they created two sets of mice with mutations in TBR1 only in layer 6. Some of the mice have no TBR1; others lack just one copy, akin to what happens in people with a TBR1 mutation. In both cases, the approach enables researchers to curtail TBR1 expression starting around birth.
The researchers then used two techniques to examine gene expression in layer 6 neurons: They sequenced all of the RNA transcripts in mashed-up tissue and then used probes to analyze the expression of nine target genes in intact tissue.
In both sets of mutants, gene expression in layer 6 neurons is more like that of layer 5 neurons than typical layer 6 neurons, the team found. This change is less pronounced in mice lacking one copy of TBR1.
TBR1 becomes active about nine days before birth in mice, but the new results suggest it is required to maintain the identity of some cell types even after birth. The finding is noteworthy because many researchers believe that a cell’s identity is locked in early on, Rubenstein says.
Several of the genes with altered expression are associated with autism. TBR1 appears to control their expression by binding to stretches of DNA that regulate the genes.
The results help flesh out the details of the relationships between autism genes — in particular, which genes affect the activity of which others, says Gill Bejerano, associate professor of developmental biology at Stanford University in California. Bejerano was not involved in the study but has previously collaborated with Rubenstein.
Thinking about therapy:
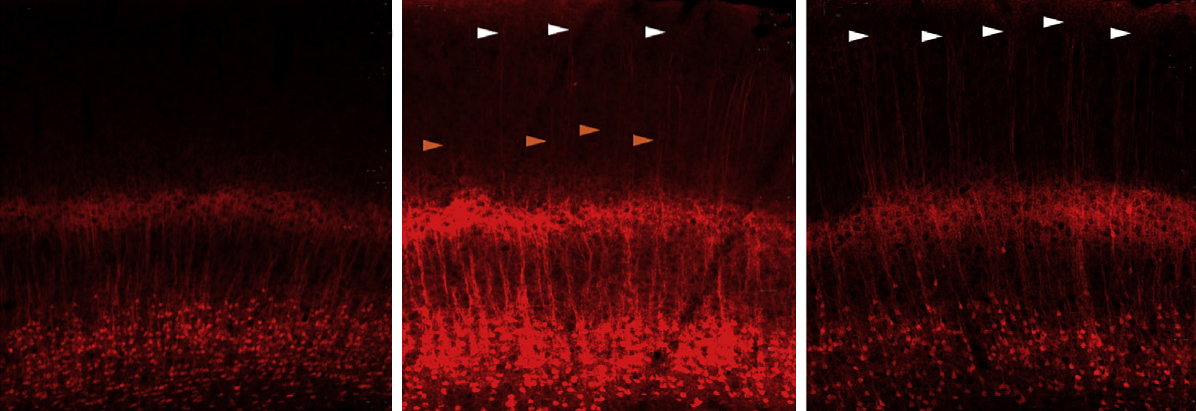
Looking in the brains of these mice, the researchers found that many of the dendrites — the signal-receiving branches of neurons — of layer 6 neurons extend to layer 1, which is typical of layer 5 neurons. “That never happens in normal layer 6 neurons,” Rubenstein says.
These dendrites also form significantly fewer connections, or synapses, with other neurons than typical layer 6 neurons do. Of the genes whose expression level changes in the mutants, four are known to affect synapse formation.
The paper is the first to link TBR1 function with dendrite connection patterns and synapse formation, says Yi-Ping Hsueh, professor of neuroscience at the Institute of Molecular Biology in Taiwan. (Hsueh has collaborated with Rubenstein but was not involved in the new work.)
The researchers used viruses to deliver functional versions of the four genes involved in synapse formation to both sets of mutant mice. One of these replacements, WNT7b, restores the number of synapses to the typical number. “That’s exciting, because it opens the possibility to think about therapy for at least the TBR1 type of autism,” Rubenstein says.
In 2014, Hsueh’s team reported that a small molecule called D-cycloserine, which boosts neuronal activity, reverses social and cognitive problems in mice lacking one copy of TBR13. It would be interesting to see whether that drug also helps alleviate problems in the layer 6 mutants, she says.
However, the overall gene-expression changes in the two types of mutants have little in common, and the mice do not have problems with social or other behaviors reminiscent of autism. For these reasons, the relevance of the findings to autism is uncertain, Hsueh says.
References:
Recommended reading
Okur-Chung neurodevelopmental syndrome; excess CSF; autistic girls

New catalog charts familial ties from autism to 90 other conditions
Explore more from The Transmitter

Karen Adolph explains how we develop our ability to move through the world
Microglia’s pruning function called into question