Astrocytes orchestrate oxytocin’s social effects in mice
The cells amplify oxytocin—and may be responsible for sex differences in social behavior, two preprints find.
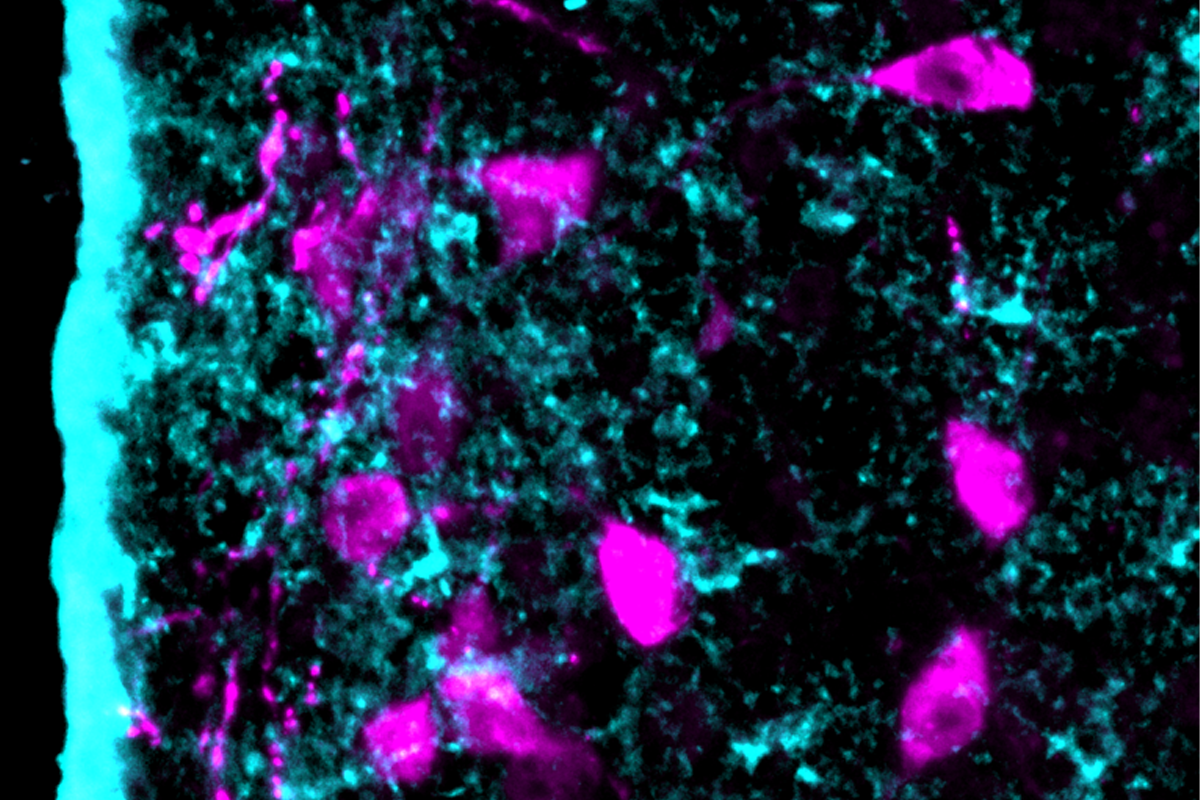
Astrocytes in the hypothalamus and the lateral septum regulate oxytocin and drive social behaviors in mice, according to two preprints posted in February. The findings offer a potential link between glial cell signaling and social difficulties associated with autism.
In the lateral septum, astrocytes respond to oxytocin to regulate social fear in mice—and these processes display striking differences between the sexes—according to one of the studies. In the hypothalamus, astrocytes propagate oxytocin production through a positive feedback loop, the other preprint found.
“There’s a growing sense that astrocytes are finely tuned to neuromodulators” and likely drive slower brain changes, including emotional states, says Ciaran Murphy-Royal, assistant professor of neuroscience at the University of Montreal, who was not involved in either study. “This work tracks well with that.”
Initial evidence that astrocytes mediate oxytocin’s effects appeared in a 2021 Nature Neuroscience paper. A subpopulation of astrocytes in the amygdala expresses oxytocin receptors and regulates anxiety in rodents, that study found. But it was unclear whether astrocytes residing in other regions perform a similar role, says Alexandre Charlet, a CNRS research director at the University of Strasbourg and an investigator on the 2021 paper.
I
Male—but not female—mice treated with the inhibitor regained sociability faster than untreated animals. By contrast, both sexes of mice lacking oxytocin receptors on astrocytes in the lateral septum displayed accelerated social fear, the study found.
The work represents “an important new vision” of glial cells modulating neuronal activity through oxytocin and other neuropeptides, says Valery Grinevich, professor of neuropeptide research in psychiatry at the University of Heidelberg, who did not contribute to the new work but was an investigator on the 2021 paper.
Male mice have fewer astrocytes in the lateral septum than female mice, and those cells sport fewer oxytocin receptors, further analysis revealed. During social avoidance, astrocytes display morphological changes only in males, the study also found.
The findings, posted on bioRxiv on 2 February, could offer insights into why women are more prone to depression and social anxiety than men, Charlet says. “Astrocytes in the lateral septum may explain, or be at the basis of, the sex differences we observe in terms of social fear.”
I
In line with that idea, Froemke and his colleagues uncovered a self-amplifying mechanism in the mouse hypothalamus. Astrocytes in that region sense oxytocin and respond by prompting neurons to produce more of it, the study found.
“There aren’t a lot of positive feedbacks in biology,” Froemke says. Harnessing this rare regulatory mechanism could indicate the importance of social relationships for reproduction and survival, he adds.
The group placed male mice in isolation for four weeks, prompting a drop in oxytocin in the paraventricular nucleus—a region of the hypothalamus that regulates social behavior. After isolation, mice take longer to cozy up to their cagemates—a behavior known as huddling—than do unseparated rodents. But isolated mice treated with intranasal oxytocin are quicker to huddle and show further oxytocin expression in the paraventricular nucleus, the study found.
Oxytocin’s effect on its own production disappeared when the team removed oxytocin receptors from astrocytes in the paraventricular nucleus, suggesting that astrocytes upregulate the neuropeptide.
T
“It’s a cool study,” says Katrina Choe, assistant professor of psychology, neuroscience and behavior at McMaster University, who was not involved in the work. “It would be interesting to know whether this [positive feedback] is an ongoing thing or whether this only happens when there are extended periods of social isolation.”
Astrocytes may have evolved to propagate oxytocin, which is otherwise quickly broken down, says study investigator Moses V. Chao, professor of cell biology, physiology and neuroscience, and psychiatry, at New York University School of Medicine. Similarly, recently discovered networks of interconnected astrocytes may also help to transmit signals to distant neurons, according to a 2025 preprint from Chao’s lab.
The findings are a “really beautiful demonstration of how this is actually happening,” says Zoe Donaldson, professor of behavioral neuroscience at the University of Colorado Boulder, who was not involved in either study. But it will be important to see whether the effects persist in larger cohorts, she says.
Chao, Froemke and their colleagues are currently repeating the experiments with more mice, including females, Froemke says. He suspects that astrocytes may regulate oxytocin differently between the sexes. “If there’s one thing that is sexually dimorphic in animals, it’s the oxytocin system,” he says.
Explore more from The Transmitter
Engrams in amygdala lean on astrocytes to solidify memories
Ramping up cortical activity in early life sparks autism-like behaviors in mice
