Gut microbes may treat social difficulties in autism mice
Consuming Lactobacillus reuteri, a gut microbe found in yogurt and breast milk, may enhance social interactions in three mouse models of autism.
Treatment with Lactobacillus reuteri, a species of gut bacteria found in yogurt and breast milk, enhances social interactions in three mouse models of autism, a new study suggests1.
The findings lend support to the idea that the gut microbiome — the population of microbes in the body — holds clues to autism therapies.
The results jibe with those from a 2016 study by the same team: In that study, the team reported that the same bacteria normalize social behavior in pups born to obese mice. They also align with a 2018 study showing that the bacteria enhance social behavior in mice with a mutation in SHANK3, a top autism gene.
“In all the models that we tried, the bacteria was effective,” says lead investigator Mauro Costa-Mattioli, professor and Cullen Foundation Endowed Chair of neuroscience at Baylor College of Medicine in Houston, Texas. “This is not a miracle that happened in my lab; I think that this is very general.”
The work hints that the bacteria elicit changes in behavior through signals sent by the vagus nerve — which connects the gut to the brain — that increase the hormone oxytocin in the brain. The findings were published today in Neuron.
The findings are an important confirmation of the bacteria’s potential as a treatment for features of autism, says Sarkis Mazmanian, professor of microbiology at the California Institute of Technology in Pasadena.
“There is still a lot to be learned,” Mazmanian says. “But the fact that they validate the results in three models may embolden the [bacteria’s] use in the clinic.”
The researchers do not recommend use of over-the-counter L. reuteri supplements to treat autism, however, because no one knows whether all strains are effective or what dose to use in people.
Social network:
Costa-Mattioli and his colleagues looked at three models of autism: mice with a mutation in both copies of the SHANK3 gene; mice exposed in utero to valproate (VPA), a seizure drug linked to autism risk in people; and an inbred strain of mice called BTBR that shows autism-like traits.
All three models have unusual microbiomes in their gut, and the SHANK3 and BTBR mice have a smaller proportion of L. reuteri in their gut than control mice do.
Starting at 3 weeks of age, the mice consumed the bacteria in their drinking water for four weeks. The researchers then assessed the mice’s social behavior.
Unlike controls, all three mouse models show a preference for an object over another mouse and spend less time interacting with another mouse than controls do.
After taking in the bacteria, however, the mice’s social behavior resembles that of controls.
The findings would be more convincing if the researchers could replicate the treatment’s effects at different ages, and in mice with mutations in other autism genes, says Yong-Hui Jiang, professor of pediatrics at Duke University in Durham, North Carolina, who was not involved in the study. “The VPA and BTBR models are complicated because the consistency of these models is still debatable,” he says.
Still, the results echo those from the 2018 study.
“They’re validating some of the stuff that we saw,” says Evan Elliott, assistant professor of molecular and behavioral neuroscience at Bar-Ilan University in Israel, who led the 2018 study. “It’s important to get similar findings from different laboratories.”
The results of the social-behavior tests could be skewed by some aspects of the mice: SHANK3 mice are sluggish, for example, and BTBR mice have a poor sense of smell. However, these traits remain after treatment, Costa-Mattioli’s team found, so they are unlikely to account for the bacteria’s effects on social behavior. The treatment did not alter the mice’s microbiomes, arguing against another mechanism for the behavioral effects.
The treatment improves social interactions even in mice that lack a microbiome. This result suggests that L. reuteri works independently of other gut microbes to influence social behavior.
Hitting a nerve:
Costa-Mattioli’s team ruled out an unusually ‘leaky gut’ as a mediator of the treatment’s effects. In this problem, common among people with autism, bacterial compounds leak from the intestines into the blood.
The researchers then cut the vagus nerve of the mutant mice and fed them L. reuteri. Without an intact vagus nerve, the mice do not respond to the treatment, suggesting that the bacteria send signals sent along this gut-brain connection.
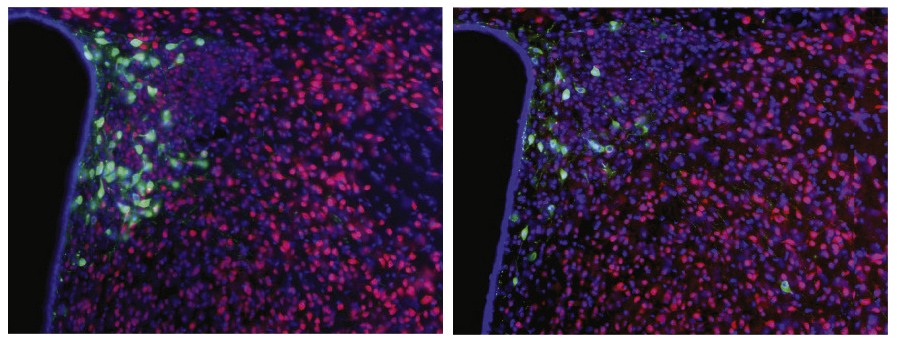
The vagus nerve connects to a brain region called the hypothalamus, which produces the hormone oxytocin. The SHANK3 mice have unusually low levels of oxytocin in the hypothalamus, the researchers found. And neurons in a reward circuit driven by oxytocin have weak connections, or synapses, in the mutants, as measured by electrical currents in brain slices.
In a 2013 study, researchers reported that feeding control mice L. reuteri increases oxytocin in the blood2. In the new study, Costa-Mattioli’s team found that L. reuteri normalizes brain oxytocin levels in the SHANK3 mice. It also boosts the strength of their neuronal connections. The bacterial treatment does not work if the mice lack oxytocin receptors in reward neurons or if they first get a drug that blocks oxytocin receptors.
Together, the results suggest that L. reuteri alters behavior by way of the vagus nerve and reward circuits that depend on oxytocin. As a next step, the team is trying to identify the bacterial compounds that activate the vagus nerve.
References:
Recommended reading

New autism committee positions itself as science-backed alternative to government group
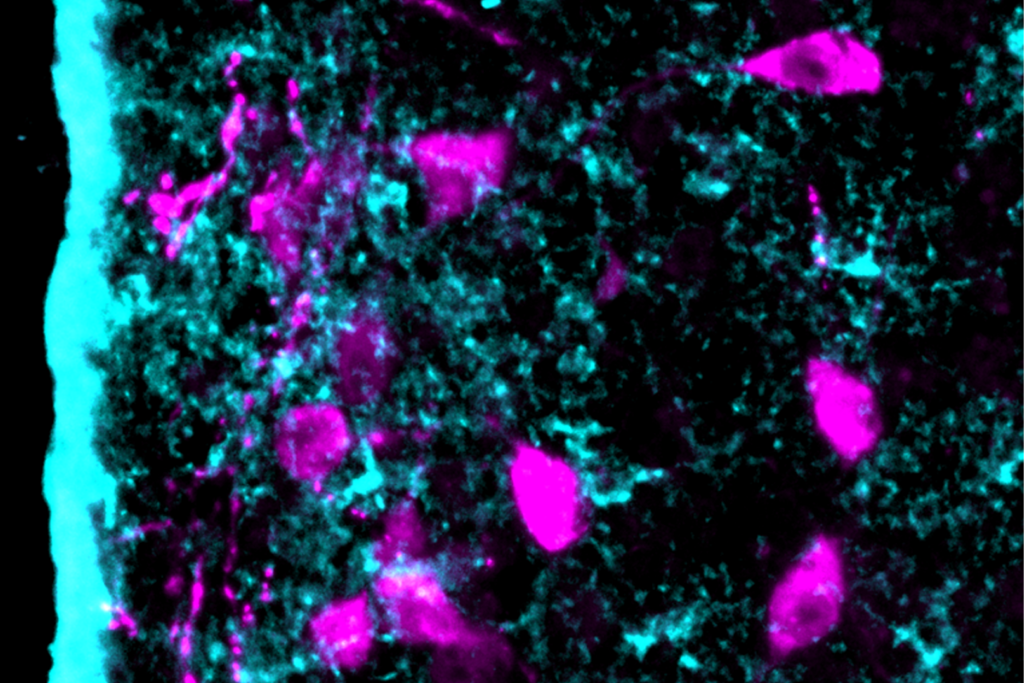
Astrocytes orchestrate oxytocin’s social effects in mice
Explore more from The Transmitter
Hippocampus builds reputation as ‘general-purpose statistical learning machine’

‘The Fox, the Shrew, and You: How Brains Evolved,’ an excerpt