Microglial overreaction to atypical neurons may drive autism
In mice and organoids lacking a neuronal protein, microglia prune synapses to excess.
When first discovered, microglia — from the Greek word meaning glue — appeared to simply prop up neurons. But that initial assessment couldn’t be further from the truth. Among other roles, they swallow invading pathogens, secrete growth factors and trim unnecessary neuronal connections.
With just one job to do, neurons are lazy in comparison, says Auguste Vadisiute, a junior research fellow at St John’s College, University of Oxford in the United Kingdom. Microglia are their support system, she says, and are critical regulators of neuronal function.
Several new strands of evidence implicate microglia as a key driver of altered brain connectivity in autism. Mice lacking the autism-linked gene SCN2A have overactive microglia and fewer neuronal connections than wildtype mice, according to a new preprint. And depleting microglia improves connectivity, the study has found.
The loss of SCN2A in neurons — the only cells that express the gene — can spur microglia into action, says study investigator Jiaxiang Wu, a postdoctoral fellow at Purdue University in West Lafayette, Indiana. The work “provides new perspectives” for treating autism by controlling the immune response, he adds.
The findings also highlight the significance of neuron-microglia interactions, says Florent Ginhoux, senior principal investigator at the Singapore Immunology Network, who was not involved in the study. “[It] reveals the important cross talk between neurons and microglia, with microglia being the driver of disease,” he says.
A
berrant microglia activity can have detrimental effects on the brain, studies from the past decade suggest. The cells’ overactivation during development — perhaps caused by an infection during pregnancy — might alter connectivity and predispose people to autism.In support of this theory, some autistic people have unusually high numbers of what appear to be active microglia, according to postmortem and live imaging studies. And the cells — which behave differently in the brains of male and female mice — may contribute to autism’s sex bias.
Yet scientists have puzzled over whether microglia play a lead role or are simply responding to altered brain conditions. So far, evidence points to the former. For example, microglia burdened with excess proteins trigger autism-like behaviors in male mice, another study found.
But some of this research has been nuanced or controversial. Bone marrow transplants — a source of microglia — reverse traits in a mouse model of Rett syndrome, a 2012 study suggested. Yet a subsequent investigation failed to replicate that finding.
In the new study, a short snippet of DNA — known as a “gene trap” — depleted SCN2A expression to 25 percent of typical levels. Mice engineered with the gene trap displayed learning and memory problems and reduced synaptic transmission.
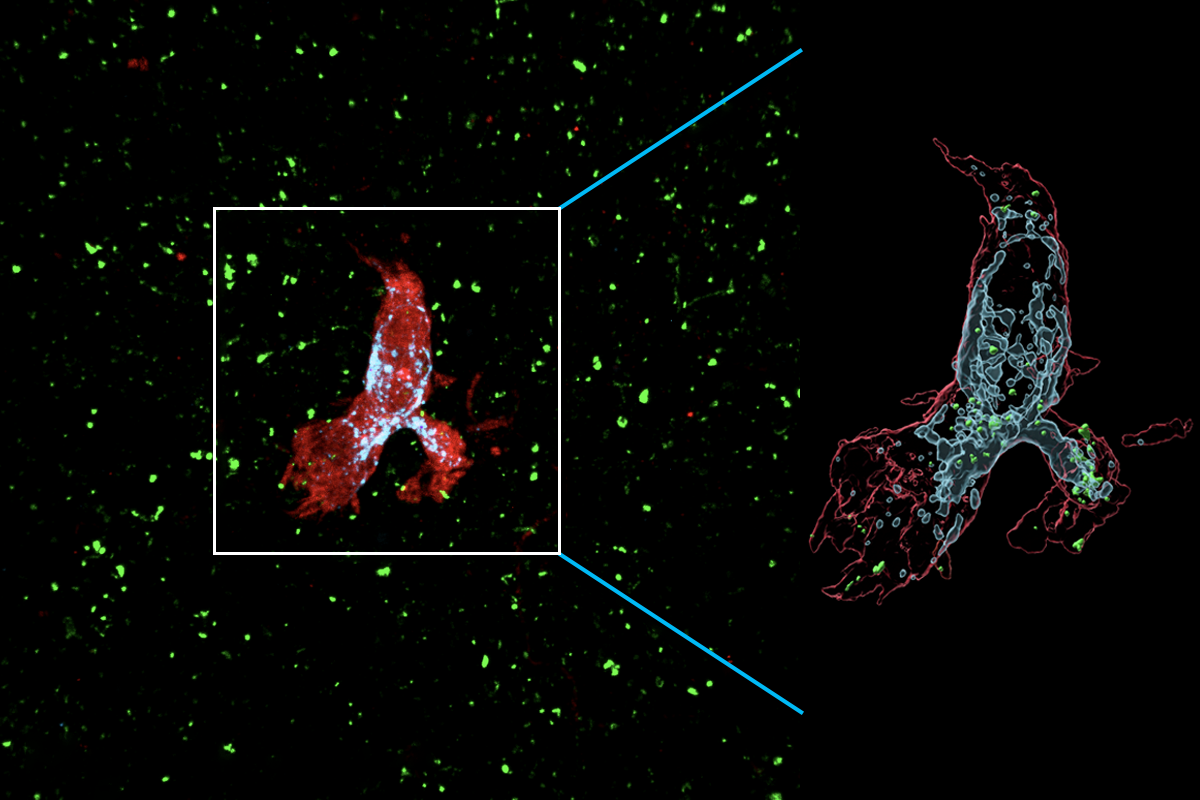
Neuronal staining in the brain’s memory hub — the hippocampus — revealed fewer dendritic spines, the tiny neuronal branches that receive signals from other neurons. Those spines were more likely to be in an immature state and formed fewer synapses than usual. These changes appeared as early as postnatal day 30, which coincides with stable levels of microglia in the mouse brain.
Microglia from SCN2A-deficient mice also produced more lysosomes — cellular sacs filled with digestive enzymes — suggesting a greater appetite for synaptic destruction. Treatment with a drug that destroys microglia partially restored synaptic transmission and spine density, the researchers found.
M
icroglia respond similarly to human neurons, the team found using cerebral organoids grown from stem cells engineered to express an autism-linked mutation in SCN2A. Microglia injected into these blobs of brain tissue became more active — and dismantled more synapses — than those added to organoids made up of unmutated neurons.The work suggests that microglial activity, rather than the loss of SCN2A — a sodium ion channel that helps neurons send electrical signals — alters connectivity in the brain, says Yang Yang, lead investigator and associate professor of medicinal chemistry and molecular pharmacology at Purdue University. The researchers posted the preprint to Research Square in September.
“It’s quite fascinating to see that they can recapitulate their key observations in the human organoid,” Ginhoux says. But they could go further, he adds, by using the model system to understand the interactions that cause microglia to react badly to atypical neurons.
The “experimental methods are state of the art,” agrees Christopher Coe, professor emeritus of biopsychology at the University of Wisconsin-Madison, who was not involved in the study. But the findings likely represent a fraction of the autistic population, he says. “There really isn’t just one type of ASD. It is likely that there won’t be just one explanation.”
Yang plans to investigate neuronal wiring in chimeric mice, in which SCN2A-mutated human neurons develop inside the mouse brain. He and his team then intend to fix the mutation to see whether microglia respond to neurons differently.
The findings from this and similar studies also suggest that microglia could be a therapeutic target, a strategy already being explored for Alzheimer’s disease. That approach, however, will require scientists to preserve the cells’ beneficial role while eliminating their detrimental effects, which won’t be easy, Yang says. “There’s a long way to go, but it’s an exciting area.”
Yang adds that he hopes more researchers will pay attention to microglia. “When we study autism, we start with a neurocentric focus,” he says. It’s past time to give the brain’s “glue” its due.
Explore more from The Transmitter


