Rett syndrome
Recent articles
Exon-skipping approach boosts levels of key Rett syndrome protein
Deleting a small region of the MECP2 gene partially restored function in neurons derived from people with Rett-associated variants.

Exon-skipping approach boosts levels of key Rett syndrome protein
Deleting a small region of the MECP2 gene partially restored function in neurons derived from people with Rett-associated variants.
Molecular changes after MECP2 loss may drive Rett syndrome traits
Knocking out the gene in adult mice triggered up- and down-regulated expression of myriad genes weeks before there were changes in neuronal function.
Molecular changes after MECP2 loss may drive Rett syndrome traits
Knocking out the gene in adult mice triggered up- and down-regulated expression of myriad genes weeks before there were changes in neuronal function.
Many autism-linked proteins influence hair-like cilia on human brain cells
The finding may help explain autism’s association with multiple co-occurring conditions that involve cilia defects.
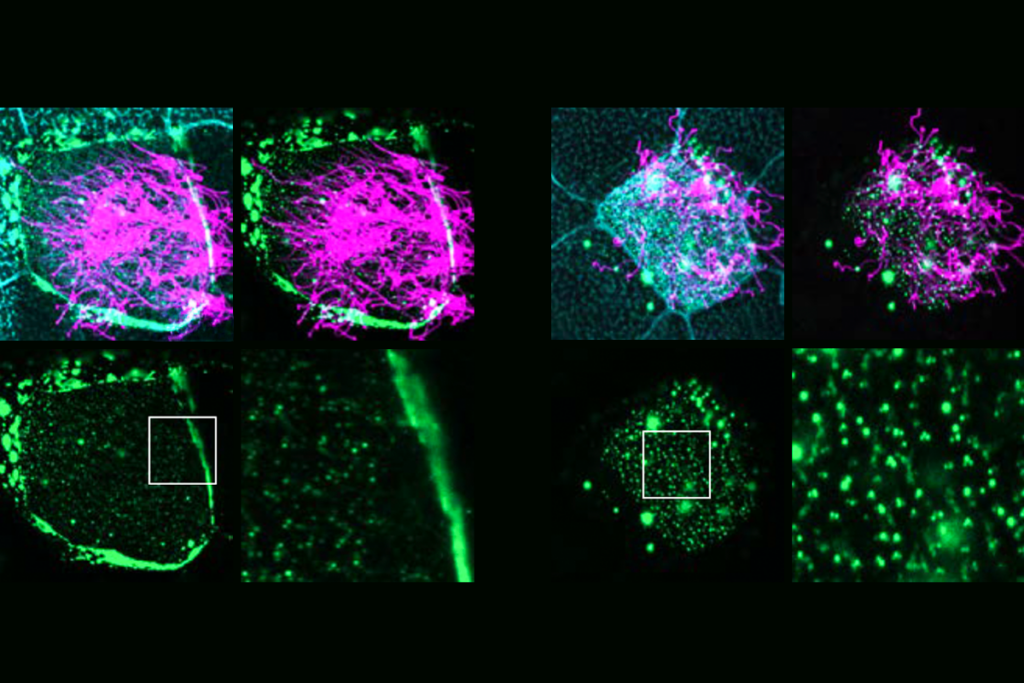
Many autism-linked proteins influence hair-like cilia on human brain cells
The finding may help explain autism’s association with multiple co-occurring conditions that involve cilia defects.
What’s next for brain-directed gene therapy after death in Neurogene trial
The incident highlights that viral vectors can trigger deadly immune responses even when delivered directly to the nervous system.
What’s next for brain-directed gene therapy after death in Neurogene trial
The incident highlights that viral vectors can trigger deadly immune responses even when delivered directly to the nervous system.
Parasite-based tool delivers MECP2 and other proteins to neurons
A method that uses a common brain parasite could help replenish the proteins deficient in Rett syndrome and other conditions.
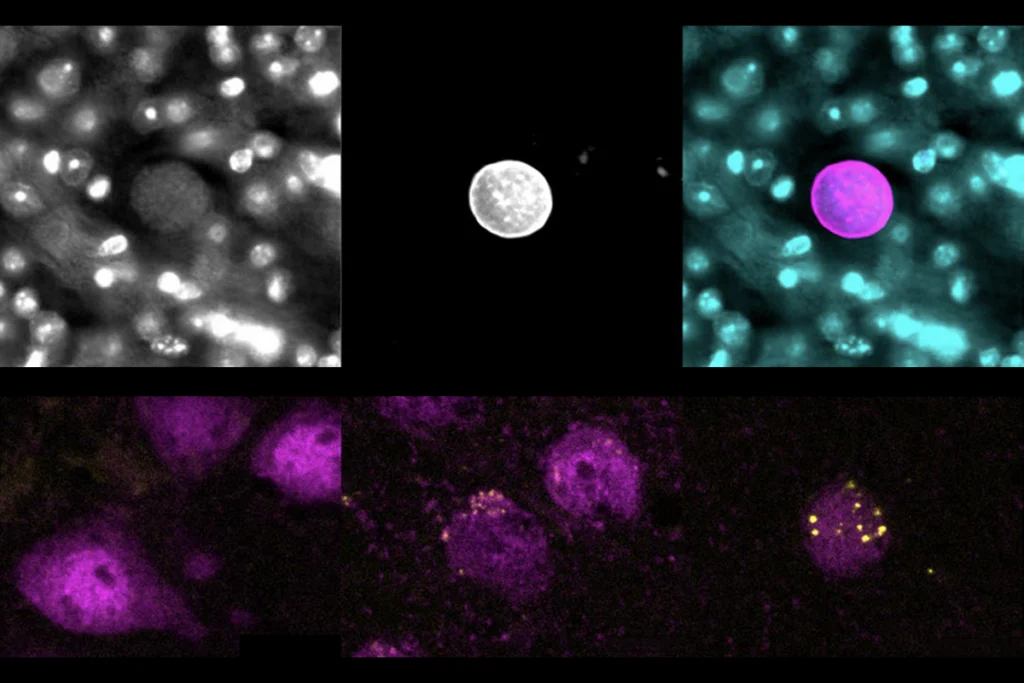
Parasite-based tool delivers MECP2 and other proteins to neurons
A method that uses a common brain parasite could help replenish the proteins deficient in Rett syndrome and other conditions.
New RNA editor boasts increased versatility, safety
The “PRECISE” technique reprograms cells in a way that, unlike DNA editors, avoids potentially permanent off-target effects.
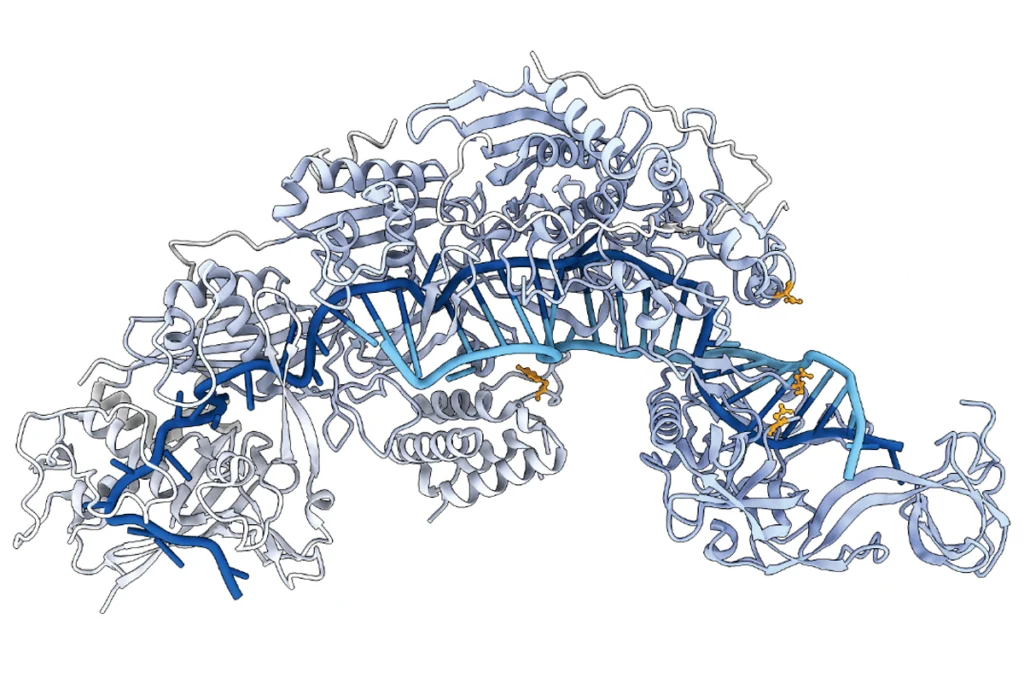
New RNA editor boasts increased versatility, safety
The “PRECISE” technique reprograms cells in a way that, unlike DNA editors, avoids potentially permanent off-target effects.
New template streamlines sharing clinical trial results
This month’s Going on Trial newsletter explores a new tool for communicating with clinical trial participants and their families, among other drug development news.
New template streamlines sharing clinical trial results
This month’s Going on Trial newsletter explores a new tool for communicating with clinical trial participants and their families, among other drug development news.
Trials test utility of EEG biomarkers for autism-related conditions
This month’s Going on Trial newsletter dives into an electroencephalography biomarker that could track the efficacy of treatments for dup15q and Angelman syndromes, among other drug development news.

Trials test utility of EEG biomarkers for autism-related conditions
This month’s Going on Trial newsletter dives into an electroencephalography biomarker that could track the efficacy of treatments for dup15q and Angelman syndromes, among other drug development news.
How organ-on-a-chip models can help drug development
This month’s Going on Trial newsletter explores how organ-on-a-chip models could smooth the transition from preclinical to clinical trials, among other drug development news.
How organ-on-a-chip models can help drug development
This month’s Going on Trial newsletter explores how organ-on-a-chip models could smooth the transition from preclinical to clinical trials, among other drug development news.
Going on Trial: Gene therapy for Rett; return to arbaclofen
This month’s newsletter looks at the early safety data from the first gene therapy trial for Rett syndrome, among other drug development news.

Going on Trial: Gene therapy for Rett; return to arbaclofen
This month’s newsletter looks at the early safety data from the first gene therapy trial for Rett syndrome, among other drug development news.
Explore more from The Transmitter
‘Unbelievably beautiful’ evidence extends Nobel Prize-winning model of vision
Orientation tuning—the ability to distinguish a horizontal line from a vertical one or something in between—originates in the visual cortex, according to new mouse synapse imaging experiments.

‘Unbelievably beautiful’ evidence extends Nobel Prize-winning model of vision
Orientation tuning—the ability to distinguish a horizontal line from a vertical one or something in between—originates in the visual cortex, according to new mouse synapse imaging experiments.
Bringing basic biology back to INSAR
As the International Society for Autism Research has grown over the past two decades, basic science has become less central, Christine Wu Nordahl says. This year, she and other meeting organizers aimed to change that.

Bringing basic biology back to INSAR
As the International Society for Autism Research has grown over the past two decades, basic science has become less central, Christine Wu Nordahl says. This year, she and other meeting organizers aimed to change that.
Every neuroscience lab needs an ethicist
The ethics issues that arise in neuroscience research are usually novel, unresolved and understudied. Embedding ethicists in labs helps scientists navigate these challenges and develop strategies in real time to prevent harm.

Every neuroscience lab needs an ethicist
The ethics issues that arise in neuroscience research are usually novel, unresolved and understudied. Embedding ethicists in labs helps scientists navigate these challenges and develop strategies in real time to prevent harm.