New clues connect altered patterns of DNA tags to autism
Methyl tags on DNA are distributed differently in postmortem brains from people with autism than in control brains, and mouse pups can inherit altered methylation from their older fathers, report two new studies.
Since the 1970s, scientists have known that autism is at least in part genetic. They are now investigating whether autism risk is also influenced by epigenetic modifications — chemical tags that bind DNA and DNA packaging proteins and influence gene expression. Changes in these chemical tags may help explain how environmental factors, such as infection during pregnancy or a parent’s age, can alter autism risk.
Two new studies reveal changes in DNA methylation, a subtype of epigenetic modification, in autismand explore theories about where such alterations originate, whether in the womb or in sperm prior to fertilization.
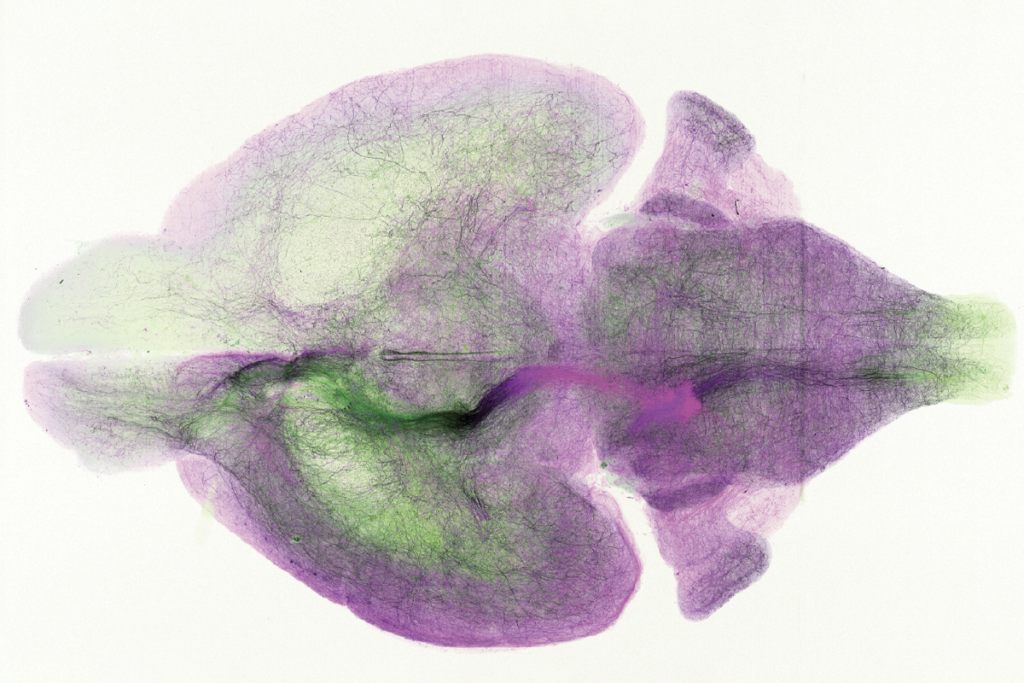
The first, published 2 September in Translational Psychiatry, shows that methyl groups are distributed differently in postmortem brains from people with autism than in control brains.
Other studies have found altered methylation patterns in postmortem brains from people with autism, as well in their blood and cheek cells. The new study finds evidence of changes in new brain regions and genes.
The researchers looked at methylation in the postmortem brains of 13 people who had autism and 12 controls. They focused on two regions in the cortex associated with atypical activity in people with autism: the prefrontal cortex and the anterior cingulate gyrus.
Methylation patterns in these two regions look dramatically more similar to each other in autism brains than in control brains. This may be because the regions do not fully differentiate in autism brains, the researchers say. This finding fits with a 2011 survey of gene expression, which similarly showed fewer differences in expression patterns between brain regions in the postmortem brains of people with autism.
The study also found intriguing methylation changes in the prefrontal cortex, which participates in reasoning and planning. Here, people with autism have decreased methylation of some immune genes, which they consequently tend to overexpress. In the brain, some immune proteins that normally fight infection have special roles, pruning synapses, or junctions between neurons, and aiding in the generation of support cells that help neurons develop.
The researchers hypothesize that infection during pregnancy — which is associated with increased risk of having a child with autism — may alter methylation of these immune genes.
Methylation might go off the rails long before pregnancy, of course. A second study, published 5 August in Molecular Psychiatry, shows that as male mice age, the methylation patterns in their sperm change and these changes are passed on to their pups.
As in mice, methylation patterns in men’s sperm change as they age, according to a July study that analyzed sperm collected from the same men overone to two decades.This is interesting because older men appear to be at elevated risk of having children with autism.
The new study measured methylation across the genomes of sperm collected from one group of mice at 3 months and another group at 12 to 14 months.They bred these mice with 3-month-old females and analyzed methylation in the pups’ brains.
Older sperm have less methylation in certain areas than younger sperm do, the study found.The pups have this same pattern, and also show altered gene expression, particularly in genes associated with early brain development. Pups born to older mice are also less likely to explore or to be startled than those born to younger mice.
It is unclear what, in any, implications these mouse findings have for people. But they do show that, in principle, age-related epigenetic changes in sperm can affect subsequent generations. The next step would be to compare methylation patterns in children who have autism with those in their fathers’ sperm.
Explore more from The Transmitter