Gene activity in human cortex shows striking sex differences
The results mark a “dramatic shift” in how neuroscientists think about sex differences, and they may help explain sex biases in certain neurodegenerative and neurodevelopmental conditions.
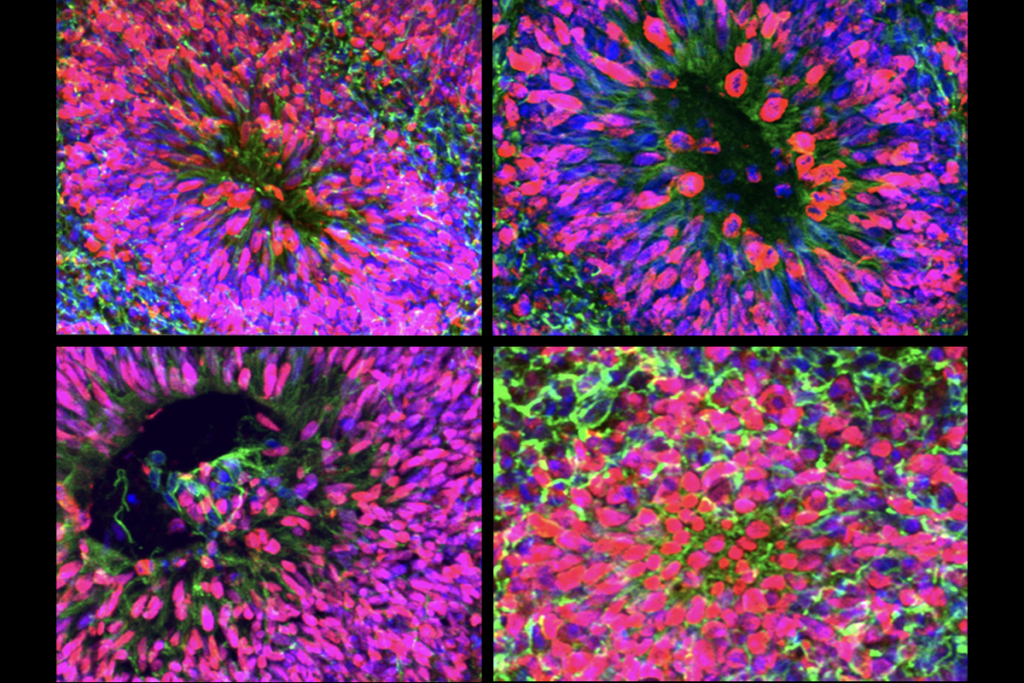
More than 3,000 genes are differently expressed in the cerebral cortex of people with XX versus XY sex chromosomes, according to a single-cell transcriptomics study published last month in Science. The differences could help explain why certain neurodegenerative and neurodevelopmental conditions affect one group more than the other, or vice versa.
The results present “a pretty dramatic shift in how we’re thinking about sex differences,” says Tomasz Nowakowski, associate professor of neurological surgery, anatomy and psychiatry, and of behavioral sciences, at the University of California, San Francisco. He was not involved in the new work but uncovered gene expression differences in prenatal developing brains last year. Previous research traced sex differences to subcortical structures, where sex hormone receptors are expressed, but “the cortex is not the part of the brain that you typically think of when you think about sex differences,” he says. “I think it’s a landmark.”
Of the thousands of genes flagged in the new study, 133 showed consistent sex differences across all brain cell types in six cortical regions sampled from postmortem brains, donated by 15 men (who all had XY sex chromosomes) and 15 women (who all had XX sex chromosomes), aged 26 to 78 years. Two of these regions—the fusiform gyrus and the inferior lateral temporo-occipital cortex—have more gray-matter volume in men, previous MRI studies suggest; two others, the caudal insula and intraparietal sulcus, have more gray matter in women; and the final two regions, the angular gyrus and the retrosplenial cortex, show no sex bias in gray-matter volume.
Intriguingly, 119 of the 133 genes are autosomal, meaning men and women should have, at least in theory, an equal dose. That makes them “ground zero for molecular sex differences in the brain,” says study investigator Armin Raznahan, chief of the Section on Developmental Neurogenomics at the U.S. National Institute of Mental Health.
M
any of the 3,000 genes identified are involved in neurodevelopmental processes and are regulated by sex steroid hormones and the X chromosome. And the sex bias in gene expression matches up with sex differences in genetic risk seen in certain brain conditions; female-biased genes show enrichment for women-specific multiple sclerosis risk genes flagged by genome-wide association studies, for example.“It’s casting kind of a wide net, but it could be capturing some of the major players” in sex bias in these conditions, says Matthew Oetjens, associate professor of developmental medicine at Geisinger College of Health Sciences, who was not involved in the study. “It doesn’t necessarily implicate a gene, but I think it’s reducing that search space.”
Across brain regions, the biggest sex differences in gene expression occur in the fusiform gyrus, which is implicated in autism because of its role in facial recognition. But the team cautions against making a one-to-one link between gene expression and behavioral differences. “When we find something at a specific biological level, that’s all we can know,” says study investigator Alex DeCasien, Stadtman Investigator in neurogenetics and head of the Computational and Evolutionary Neurogenomics Unit at the U.S. National Institute on Aging. “We can’t make assumptions about the underlying drivers or the downstream consequences.”
Adding to the complexity, some male-biased and female-biased genes overlap in the same cell compartments and biological processes, Raznahan says, which means “sex differences of one level could be working to counterbalance each other, so you don’t see sex differences at the next level of organization.”
For example, no one cell type emerged as having the biggest differences in gene expression between XX and XY people. Instead, the results changed based on the statistical test used, the researchers say.
Genes found on sex chromosomes show the largest differences in expression, which lines up with Nowakowski’s team’s findings last year. That’s “kind of an acid test,” Raznahan says. “There seem to be pretty consistent lifespan differences in expression of those genes that are dosage sensitive.”
Typically in people with XX chromosomes, one X chromosome is silenced, but some genes can escape that silencing and are expressed at increased levels, DeCasien says. “We were able to actually nominate some genes for the first time,” they add, because previous studies looked only in blood or cell cultures. “Those genes already represent a hot spot for sex differences at the molecular level,” and some are implicated in neurodevelopmental conditions.
Sex explains a relatively small percentage—just 0.3 percent—of the variance in gene expression across the study’s samples, whereas at a population level it “can be one of the biggest explainers of variation” in a condition’s prevalence, Oetjens says; autism, for example, is diagnosed about four times more often in boys than in girls.
The researchers say they plan to further investigate the function of differently expressed genes, including using organoids cultivated from people with atypical sex chromosomes, such as those with Klinefelter syndrome and trisomy X syndrome. But DeCasien says they also “have a goal of moving beyond sex as an investigative term.” Currently, the concept of sex is used in research as a proxy for sex chromosomes and sex hormones, but also for environmental input, which has a gender bias but doesn’t necessarily align with the other two variables. One way to untangle these different factors would be, for example, to map the relationship between circulating hormones and gene expression in the brain at different points in life, DeCasien says.
The important thing is for the field to hold in mind the complexity of sex- and gender-driven effects, Raznahan says. “I think that really is crucial.”
Explore more from The Transmitter

Among brain changes studied in autism, spotlight shifts to subcortex