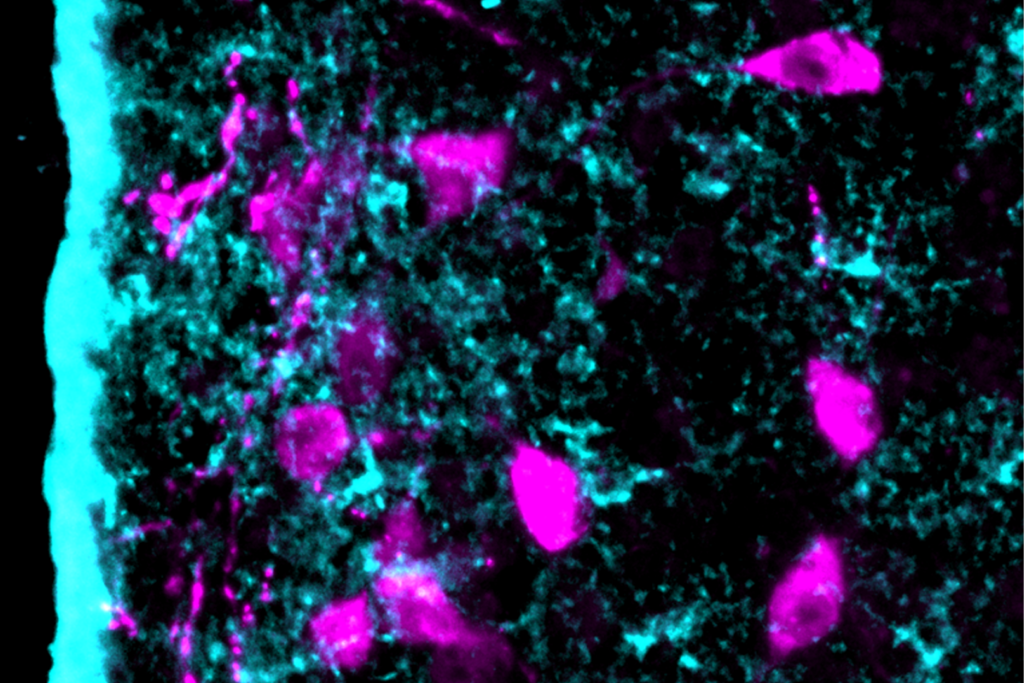
Mutations in SHANK3, a leading autism candidate gene, disrupt a brain circuit linked to social behavior, a new study in mice finds.
Discovering how autism traits relate to specific neural circuits could lead to circuit-based therapies, says Camilla Bellone, associate professor of basic neurosciences at the University of Geneva in Switzerland, who did not take part in this research.
The social-interaction difficulties that characterize autism arise from altered communication between different parts of the brain, past research suggests — with some findings homing in on particular circuits: Overactivity between the prefrontal cortex, which regulates social activity, and the basolateral amygdala, which plays a role in learning about rewarding or unwanted outcomes, for example, appears to decrease sociability in wildtype mice.
In the new study, the researchers used genetically modified viruses to delete SHANK3, which encodes a key protein for neuronal communication, from this circuit in adult mice.
“Conventional mouse genetic studies in neuropsychiatric disorders mostly focus on the whole brain, brain regions or cell types,” says lead investigator Il Hwan Kim, assistant professor of anatomy and neurobiology at the University of Tennessee Health Science Center in Memphis. “We attempted to investigate a risk gene’s effects on a single neural circuit.”
N
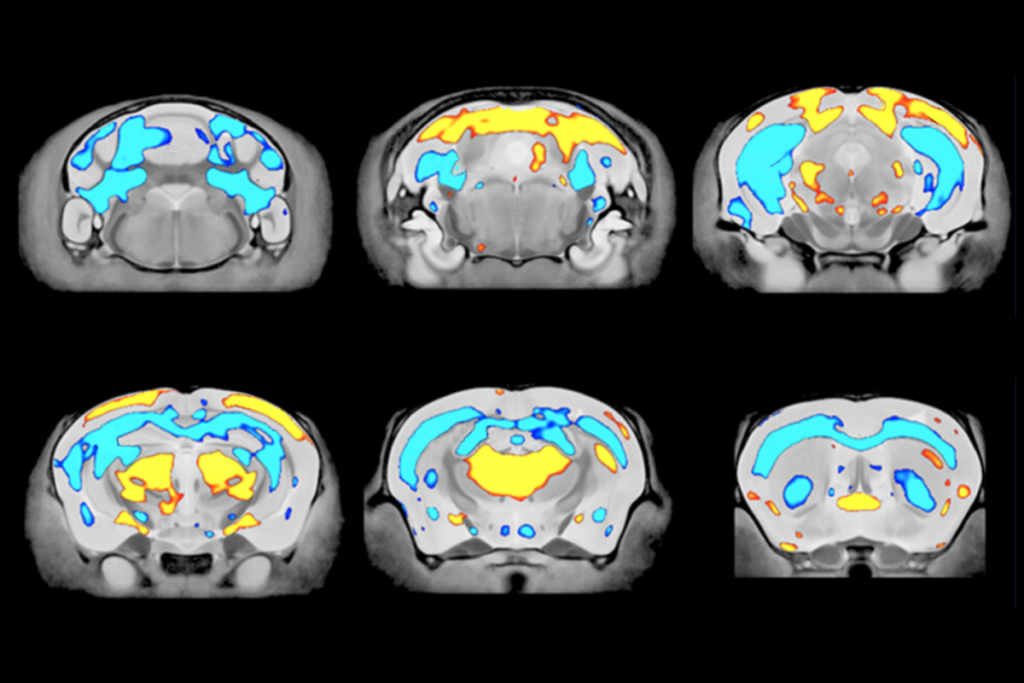
eurons in the prefrontal cortex-basolateral amygdala circuit in the modified SHANK3 mice sprouted shorter, denser dendrites compared with controls. The neurons also displayed increased excitatory and decreased inhibitory activity, with the mice showing elevated neural activity in the circuit, as well as greater variability in the strength of signals in the circuit. Autistic people’s brains show similar overactivity in this circuit, according to resting-state functional magnetic resonance images from the Autism Brain Imaging Data Exchange dataset.Some of the neurons in the circuit specifically respond to social cues, the scientists discovered when they analyzed the neuronal activity of wildtype mice. In modified male mice, these socially tuned neurons became less responsive to social interactions. The modified male mice also showed decreased sociability in the form of reduced sniffing of other mice.
The modified female mice did not display any significant social differences, consistent with findings that suggest autism manifests differently in girls versus boys, the researchers note. This “intriguing” result should prompt future studies “to mechanistically investigate these differences between male and female [mice],” Bellone says.
In wildtype mice, overactivating the circuit in question using optogenetics partially lowered their sociability, the team also found. On the other hand, inhibiting this circuit in the SHANK3 mice partially rescued their social behavior. The scientists detailed their findings earlier this month in Cell Reports.
“This study advances our understanding of the complex neural circuitry underlying social deficits in model organisms like rodents, and how their activity can be disrupted by a prominent and well-characterized autism-related mutation,” says Alessandro Gozzi, senior scientist at the Italian Institute of Technology in Rovereto, Italy, who was not involved in the study. “It would be very interesting to understand whether other mutations may similarly impact the circuit investigated by the authors.”
I
n the future, the scientists plan to expand their scope to investigate the neurons this circuit influences in the basolateral amygdala. In addition, “we hope to find how the prefrontal cortex-basolateral amygdala circuit affects a larger network of brain regions that are known to be involved during social behavior,” Kim says.Mutations in the schizophrenia risk gene ARP2/3 within the same neural circuit trigger similarly reduced social behavior, Kim says he and his colleagues previously found — highlighting “a shared hallmark symptom of autism and schizophrenia.” Even though mutations in SHANK3 and ARP2/3 result in different changes in the shape of neurons in lab animals, their neural activity changed in a similar manner, he says.
Together, these past and new findings “may explain how different risk genes that are associated with different neuropsychiatric disorders can drive similar behavioral symptoms,” Kim says. “In the future, we can shift our focus from a conceptual and broad frame of a neuropsychiatric disorder to specific targeting of a behavioral abnormality.”
Bellone notes that this work involved adult mice instead of young mice whose brains are still developing. “It will be interesting to know in the future how the deletion of the gene during critical periods may affect circuit development,” she says.
And many other brain regions and circuits govern social behavior, Bellone adds, suggesting that similar investigations of these other circuits could potentially “be translated in a circuit-based therapeutic intervention in humans.”